
Cómo usar Colobreathe 1.662.500 UI polvo para inhalacion, capsulas duras
Traducción generada por IA
Este contenido ha sido traducido automáticamente y se ofrece solo con fines informativos. No sustituye la consulta con un profesional sanitario.
Ver originalContenido del prospecto
Introducción
Prospecto: información para el usuario
Colobreathe 1.662.500 UI polvo para inhalación, cápsulas duras
colistimetato de sodio
Lea todo el prospecto detenidamente antes de empezar a usareste medicamento, porque contiene información importante para usted.
- Conserve este prospecto, ya que puede tener que volver a leerlo.
- Si tiene alguna duda, consulte a su médico o farmacéutico.
- Este medicamento se le ha recetado solamente a usted, y no debe dárselo a otras personas tengen presenten los mismos síntomas que usted, ya que puede perjudicarles.
- Si experimenta efectos adversos, consulte a su médico o farmacéutico, incluso si se trata de efectos adversos que no aparecen en este prospecto. Ver sección 4.
Contenido del prospecto
- Qué es Colobreathe y para qué se utiliza
- Qué necesita saber antes de empezar a usar Colobreathe
- Cómo usar Colobreathe
- Posibles efectos adversos
- Conservación de Colobreathe
- Contenido del envase e información adicional
1. Qué es Colobreathe y para qué se utiliza
Colobreathe contiene colistimetato de sodio, un tipo de antibiótico llamado polimixina.
Colobreathe se utiliza para controlar infecciones pulmonares persistentes causadas por la bacteria Pseudomonas aeruginosaen pacientes adultos y niños de 6 años de edad y mayores con fibrosis quística. Pseudomonas aeruginosaes una bacteria muy común que infecta a casi todos los pacientes con fibrosis quística en algún momento de sus vidas. Algunas personas contraen esta infección mientras son muy jóvenes, pero para otros ocurre mucho más tarde. Si esta infección no se controla adecuadamente, producirá daño a los pulmones.
Cómo actúa
Colobreathe actúa destruyendo la membrana celular bacteriana, con un efecto letal sobre estas bacterias.
2. Qué necesita saber antes de empezar a usar Colobreathe
No use Colobreathe:
- si usted/su hijo es alérgico al colistimetato de sodio, al sulfato de colistina o las polimixinas.
Advertencias y precauciones
Consulte a su médico o farmacéutico antes de empezar a usar Colobreathe.
Informe a su médico si usted/su hijoha padecido alguna vez cualquiera de las afecciones siguientes:
- ha reaccionado mal anteriormente a la inhalación de medicamentos en polvo seco, a menos que esto ya se haya discutido con su médico.
- ya tiene una enfermedad muscular conocida como miastenia gravis o la enfermedad hereditaria, porfiria
- sangre en el esputo (la sustancia que expulsa al toser)
Después de cada inhalación de Colobreathe, se debe enjuagar la boca con agua. El enjuague no debe ser ingerido. El enjuague puede reducir el riesgo de desarrollar sobreinfecciones orales por hongos durante el tratamiento, y también puede reducir el sabor desagradable asociado al colistimetato de sodio.
Cuando usted/su hijo comience a usar Colobreathe, usted/su hijo puede encontrar que tiene tos, dificultad al respirar, opresión en el pecho o sibilancias (sonido silbante que se produce al respirar). El número de estos efectos adversos se puede reducir a medida que continúe el uso del inhalador o puede que su médico le recete un broncodilatador para utilizar antes o después de tomar Colobreathe. Si cualquiera de estos efectos llega a ser un problema, por favor póngase en contacto con su médico quien pueda modificar su tratamiento.
Si usted/su hijo presenta cualquier problema en los riñones o nervios, se deben extremar las precauciones a la hora de administrar Colobreathe, aunque su médico ya estará al corriente de ello.
Si usted/su hijo requiere otras formas de colistimetato, ya sea por inyección o por nebulización, se deben extremar las precauciones, aunque su médico ya estará al corriente de ello.
Niños
No administrar Colobreathe a niños menores de 6 años de edad, ya que no es apropiado para ellos.
Otros medicamentos yColobreathe
Informe a su médico si usted/su hijo está tomando, ha tomado recientemente o podría tener que tomar cualquier otro medicamento, y en particular:
- si a usted/su hijo está tomando antibióticos aminoglucósidos, utilizados para tratar infecciones, se deben extremar las precauciones;
- si usted/su hijo padece miastenia grave y está tomando antibióticos del tipo de los macrólidos como la azitromicina y la claritromicina, o fluoroquinolonas como el norfloxacino y el ciprofloxacino. Tomar éstos al mismo tiempo que Colobreathe puede producir problemas de debilidad muscular;
- si a usted/su hijo está tomando colistimetato por inyección o nebulización, se deben extremar las precauciones;
- si usted/su hijo requiere una anestesia general, se deben extremar las precauciones.
Embarazo y lactancia
Si está embarazada o en periodo de lactancia, o cree que podría estar embarazada o tiene intención de quedarse embarazada, consulte a su médico o farmacéutico antes de utilizar este medicamento.
No hay información sobre la seguridad de Colobreathe en mujeres embarazadas. Su médico
debe aconsejarle antes de usar Colobreathe acerca de si los beneficios del medicamento superan los riesgos.
El colistimetato de sodio puede pasar a la leche materna. Hable sobre el uso de Colobreathe con su médico.
Conducción y uso de máquinas
Es posible que mientras usa Colobreathe pueda experimentar mareo, confusión o tener problemas de visión. No conduzca ni maneje máquinas hasta que los síntomas hayan desaparecido.
Colobreathe contiene sodio
Este medicamento contiene menos de 1 mmol de sodio (23 mg) por cápsula; esto es, esencialmente “exento de sodio”.
3. Cómo usar Colobreathe
Siga exactamente las instrucciones de administración del medicamento contenidas en este prospecto o las indicadas por su médico. En caso de duda, pregunte a su médico si usted/su hijo tiene dudas.
La primera dosis se debe administrar bajo supervisión médica.
La dosis recomendada es
Adultos y niños de 6 años de edad y mayores
- El contenido de una cápsula de Colobreathe debe ser inhalado dos veces al día usando el inhalador Turbospin.
- Debe haber un espacio de 12 horas entre dosis.
El orden en que se deben tomar o realizar otros tratamientos
Si usted/su hijo está tomando otros tratamientos para la fibrosis quística, usted/su hijo debe tomarlos en el siguiente orden:
- Broncodilatadores inhalados
- Fisioterapia respiratoria
- Otros medicamentos inhalados
- Luego Colobreathe
Usted/su hijo debe confirmar el orden de sus tratamientos con su médico.

Forma de administración
Colobreathe se inhala hacia los pulmones en forma de polvo desde la cápsula usando el inhalador manual llamado Turbospin. Colobreathe solo puede ser administrado utilizando este dispositivo.
No ingerir las cápsulas de Colobreathe.
Para inhalar Colobreathe desde la cápsula mediante el inhalador Turbospin, siga el procedimiento descrito a continuación. Su médico, farmacéutico o enfermera debe mostrarle a usted/a su hijo cómo inhalar el medicamento cuando usted/su hijo inician el tratamiento:
Usar Colobreathe con el inhalador Turbospin
Preparación del Turbospin
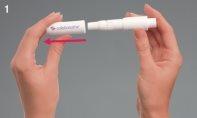
- Retire la capucha. Sale tirando suavemente de ella.

- Desenrosque la boquilla, dejando al descubierto la cámara del inhalador Turbospin.

- Extraiga una sola cápsula del blíster. Una vez que haya extraído la cápsula, debe usarla de inmediato.

- Inserte suavemente la cápsula en la cámara con el extremo más ancho primero. No hay que hacer fuerza.

- Ahora vuelva a colocar la boquilla enroscándola en su lugar.
Perforar la cápsula e inhalar el medicamento

- Para perforar la cápsula:
- Sujete el inhalador con la boquilla hacia arriba, empuje el pistón suavemente hacia arriba hasta llegar a la línea visible; notará resistencia en este punto y esto fijará la cápsula en su lugar, lista para ser perforada. Mantenga esa posición antes de proceder a la perforación.
- Ahora, con la cápsula fijada en su lugar, siga empujando el pistón hasta llegar al tope y entonces suéltelo.
- La cápsula ahora está perforada y el contenido se puede inhalar.
- Noperfore la cápsula más de una vez. Quizá vea un poco de polvo saliendo de la cámara de la cápsula después de perforarla, lo cual es normal

- Espire lentamente. Coloque la boquilla entre los labios y los dientes. Asegúrese de crear un cierre hermético entre los labios y la boquilla. Tenga cuidado de no tapar las aberturas de aire con los dedos o la boca durante la inhalación.
- Entonces, inspire lenta y profundamente por la boca a un ritmo suficiente que le permita oír o notar que la cápsula gira.
- Retire el inhalador Turbospin de la boca y aguante la respiración durante unos 10 segundos, o durante el tiempo que se sienta cómodo, luego expulse el aire lentamente.
- Si no oye girar la cápsula, puede que se haya quedado atascada en el compartimento. Si esto sucede, puede soltarla dando golpecitos suavemente a la cámara del inhalador. No intente soltar la cápsula presionando repetidamente el pistón. Si la cápsula no se puede soltar y el polvo no se puede inhalar, deshágase de la cápsula rota y de cualquier resto de polvo que quede en ella y utilice otra.
- Inhale el medicamento otra vez repitiendo los pasos 7 y 8 para asegurarse de que haya vaciado la cápsula.
- Puede verificar si la cápsula está vacía desenroscando la boquilla y comprobando la cápsula. Si no está vacía, repita los pasos 7, 8 y 9 hasta que haya inhalado todo el contenido.
- Cuando haya inhalado todo el contenido, enjuáguese bien la boca con agua y luego escúpala
Retirar la cápsula vacía del Turbospin
- Cuando la cápsula esté vacía, desenrosque la boquilla, luego extraiga y deseche la cápsula vacía.
Información adicional
Cuando usted respira lentamente, aspira aire a través del cuerpo del inhalador Turbospin dentro de la cámara de la cápsula. Las diminutas partículas de medicamento de la cápsula son recogidas por el flujo de aire y transportadas por sus vías respiratorias a los pulmones.
Ocasionalmente, trozos muy pequeños de la envoltura de la cápsula pueden entrar en la boca o las vías respiratorias.
- Si esto sucede, puede que note estos trozos en la lengua o las vías respiratorias.
- La envoltura de la cápsula está hecha de gelatina, que es inocua para los seres humanos si se traga o se inhala.
- Las probabilidades de que la cápsula se rompa en trozos aumentan si la cápsula se perfora más de una vez durante el paso 6.
Limpieza del dispositivo Turbospin
Limpie el inhalador Turbospin después de cada dosis mediante el siguiente procedimiento:
- Presione el pistón a fondo un par de veces mientras mantiene la cámara boca abajo.
- Limpie la cámara con un paño o bastoncillo de algodón. No utilice agua.
- Enrosque la boquilla firmemente de nuevo en su lugar, ponga el tapón y el inhalador estará listo para ser utilizado en la siguiente dosis.
Si usted/su hijo usa más Colobreathe del que debe, o si ha ingerido accidentalmente la cápsula,póngase en contacto con su médico inmediatamente para obtener ayuda.
Si usted/su hijo olvidó usar Colobreathe
Si usted/su hijo olvida administrarse una dosis de Colobreathe, entonces usted/su hijo se debe administrar la dosis que olvidó tan pronto como usted/su hijo lo recuerde. Usted/su hijo no se debe administrar 2 dosis en menos de 12 horas. Continúe a partir de ahí tal como se le ha indicado.
Si usted/su hijo interrumpe el tratamiento con Colobreathe
No interrumpa su tratamiento prematuramente a menos que su médico le diga que puede hacerlo. Su médico decidirá cuánto tiempo debe durar su tratamiento/el tratamiento de su hijo.
Si usted/su hijo tiene cualquier otra duda sobre el uso de este medicamento, pregunte a su médico.
4. Posibles efectos adversos
Al igual que todos los medicamentos, este medicamento puede producir efectos adversos, aunque no todas las personas los sufran.
Reacciones alérgicas
Una reacción alérgica con Colobreathe es posible (por lo general las reacciones alérgicas graves pueden causar erupciones cutáneas, hinchazón de la cara, lengua y cuello, dificultad para respirar debido al estrechamiento de las vías respiratorias y pérdida de conciencia). Si usted/su hijo experimenta signos de una reacción alérgica, debe buscar atención médica urgente.
Otros posibles efectos adversos
Usted/su hijo puede tener un sabor desagradable en la boca después de la inhalación de Colobreathe.
Muy frecuente (puede afectar a más de 1 persona de cada 10):
- Dificultad para respirar
- Tos, irritación de garganta
- Voz ronca o débil, o incluso pérdida de la voz
- Sabor desagradable
Frecuente (puede afectar hasta 1 persona de cada 10):
- Dolor de cabeza
- Tintineo o zumbidos en el oído, problemas con el equilibrio.
- Tos con sangre, sibilancias (sonido silbante que se produce al respirar), molestias en el pecho, asma, tos productiva (tos que produce moco), infección de los pulmones, crepitaciones en los pulmones (el médico oye esto cuando escucha los pulmones con un estetoscopio)
- Vómitos,náuseas
- Cambios en su función pulmonar (hallados en las pruebas)
- Dolor en las articulaciones
- Falta de energía, cansancio
- Aumento de la temperatura
Poco frecuente (puede afectar hasta 1 persona de cada 100):
- Reacciones alérgicas (hipersensibilidad); los signos pueden incluir erupción y picazón
- Fluctuación de peso, apetito disminuido
- Ansiedad
- Ataques
- Tendencia al sueño
- Obstrucción en los oídos
- Dolor en el pecho
- Dificultad para respirar
- Hemorragias nasales, catarro (moco en la nariz, lo que puede hacer que se sienta bloqueado) tos con moco espeso verde, dolor en la garganta y los senos paranasales
- Ruidos extraños en el pecho (su médico oiría esto al escuchar los pulmones con un estetoscopio)
- Diarrea, gases
- Producción excesiva de saliva
- Dolor dental
- Proteína en la orina (hallada en las pruebas)
- Sed
Los efectos adversos descritos anteriormente se han observado en personas de todas las edades con una frecuencia similar.
Comunicación de efectos adversos
Si experimenta cualquier tipo de efecto adverso, consulte a su médico o farmacéutico, incluso si se trata de posibles efectos adversos que no aparecen en este prospecto. También puede comunicarlos directamente a través del sistema nacional de notificación incluido en el Apéndice V. Mediante la comunicación de efectos adversos usted puede contribuir a proporcionar más información sobre la seguridad de este medicamento.
5. Conservación de Colobreathe
Mantener este medicamento fuera de la vista y del alcance de los niños.
No utilice este medicamento después de la fecha de caducidad que aparece en el embalaje exterior y en el blíster después de EXP. La fecha de caducidad es el último día del mes que se indica.
No conservar Colobreathe a temperatura superior a 25oC.
Conservar en el embalaje original hasta inmediatamente antes de su uso para protegerlo de la humedad.
Si usted/su hijo despega el papel metálico accidentalmente y alguna cápsula queda expuesta, por favor, deseche esa cápsula.
Deseche el inhalador Turbospin después de la finalización de un envase de tratamiento.
Los medicamentos no se deben tirar por los desagües ni a la basura. Pregunte a su farmacéutico cómo deshacerse de los envases y de los medicamentos que ya no necesita. De esta forma ayudará a proteger el medio ambiente.
6. Contenido del envase e información adicional
Composición de Colobreathe
El principio activo es colistimetato de sodio. Cada cápsula contiene 1.662.500 UI (aproximadamente equivalente a 125 mg) de colistimetato de sodio.
Los demás componentes son:
Cubierta de la cápsula
Gelatina
Polietilenglicol
Lauril sulfato de sodio
Agua purificada
Aspecto del producto y contenido del envase
Colobreathe polvo para inhalación, cápsula dura (polvo para inhalación) se suministra en pequeñas cápsulas de gelatina duras y transparentes que contienen un polvo blanco fino.
El Turbospin es un inhalador de polvo seco impulsado por el flujo inspiratorio, fabricado en polipropileno y acero inoxidable.
Las cápsulas están envasadas en blísteres, que se suministran en cajas que contienen:
- 56 cápsulas duras y un dispositivo inhalador de polvo Turbospin, suficientes para 4 semanas de uso.
- 8 cápsulas duras y un dispositivo inhalador de polvo Turbospin, suficientes para 4 días de uso.
Puede que solamente estén comercializados algunos tamaños de envases.
Titular de la autorización de comercialización
Essential Pharma Limited,
Vision Exchange Building,
Triq it-Territorjals, Zone 1,
Central Business District,
Birkirkara, CBD 1070,
Malta
Responsable de la fabricación
Teva Pharmaceuticals Europe BV
Swensweg 5
2031 GA Haarlem
Países Bajos.
Millmount Healthcare Limited
Block 7, City North Business Campus
Stamullen
Co Meath
K32 YD60
Irlanda
Merckle GmbH
Ludwig-Merckle-Str-3
89143 Blaubeuren
Alemania
Laboratorios Liconsa, S.A.
Avda. Miralcampo, 7, Pol. Ind.
Miralcampo
19200 Azuqueca de Henares (Guadalajara)
España
Fecha dela última revisión de este prospecto: febrero 2024.
La información detallada de este medicamento está disponible en la página web de la Agencia Europea de Medicamentos: http://www.ema.europa.eu. También existen enlaces a otras páginas web sobre enfermedades raras y medicamentos huérfanos.
-

- País de registro
- Forma farmacéuticaINHALACIÓN PULMONAR, 1.662.500 UI
- Código ATCJ01XB01
- Principio activo
- Requiere recetaSí
- Fabricante
- Esta información es de carácter general y no sustituye la consulta con un profesional sanitario.
- Alternativas a Colobreathe 1.662.500 UI polvo para inhalacion, capsulas durasForma farmacéutica: INHALACIÓN PULMONAR, 1 MIUPrincipio activo: colistinaFabricante: Pari Pharma GmbhRequiere recetaForma farmacéutica: INHALACIÓN PULMONAR, 2 MIUPrincipio activo: colistinaFabricante: Pari Pharma GmbhRequiere recetaForma farmacéutica: INYECTABLE, 1MUIPrincipio activo: colistinaFabricante: Accord Healthcare S.L.U.Requiere receta
Médicos online para Colobreathe 1.662.500 UI polvo para inhalacion, capsulas duras
Comenta la dosis, los posibles efectos secundarios, interacciones, contraindicaciones o la revisión de receta de Colobreathe 1.662.500 UI polvo para inhalacion, capsulas duras, sujeto a valoración médica y a la normativa local.
Obtén una receta de Colobreathe 1.662.500 UI polvo para inhalacion, capsulas duras online
Rellena un formulario de 2 minutos
Cuéntanos tus síntomas, historial y el medicamento que necesitas.
Elige un médico o nosotros te asignamos uno
Escoge un especialista o te conectamos con el próximo médico disponible.
El médico revisa tu caso
Normalmente en 30 minutos. Puede hacerte preguntas adicionales por chat.
Recógela en cualquier farmacia
Receta electrónica enviada a tu correo, válida en toda España.
Preguntas frecuentes
Colobreathe 1.662.500 UI polvo para inhalacion, capsulas duras requires receta en España. Puedes confirmarlo con un médico online según tu caso.
El principio activo de Colobreathe 1.662.500 UI polvo para inhalacion, capsulas duras es colistina. Esta información ayuda a identificar medicamentos con la misma composición pero con diferentes nombres comerciales.
Colobreathe 1.662.500 UI polvo para inhalacion, capsulas duras es fabricado por Essential Pharma Limited. La marca y el envase pueden variar según el distribuidor.
Los Médicos de familia, Psiquiatras, Dermatólogos, Cardiólogos, Endocrinólogos, Gastroenterólogos, Neumólogos, Nefrólogos, Reumatólogos, Hematólogos, Infectólogos, Alergólogos, Geriatras, Pediatras, Oncólogos pueden valorar la prescripción de Colobreathe 1.662.500 UI polvo para inhalacion, capsulas duras cuando esté clínicamente indicado. Puedes reservar una videoconsulta para comentar tu caso y las opciones de tratamiento.
España cuenta con una infraestructura sanitaria bien desarrollada en ciudades principales como Madrid, Barcelona, Valencia y Sevilla. Las farmacias están ampliamente disponibles y operan bajo estrictas regulaciones, garantizando el acceso a medicamentos con receta.
Puedes comprar Colobreathe 1.662.500 UI polvo para inhalacion, capsulas duras en Madrid, Barcelona, Sevilla o Valencia en cualquier farmacia local con una receta válida.
Para obtener una receta, puedes utilizar Oladoctor:
Otros medicamentos con el mismo principio activo (colistina) incluyen COLFINAIR 1 MILLON DE UI POLVO PARA SOLUCION PARA INHALACION POR NEBULIZADOR, COLFINAIR 2 MILLONES DE UI POLVO PARA SOLUCION PARA INHALACION POR NEBULIZADOR, COLISTIMETATO DE SODIO ACCORD 1 MILLON DE UI POLVO PARA SOLUCION INYECTABLE Y PARA PERFUSION EFG. Pueden tener diferentes nombres comerciales o formulaciones, pero contienen el mismo componente terapéutico. Consulta siempre con un médico antes de cambiar o iniciar un nuevo medicamento.






