
RIZATRIPTAN FLAS CINFA 10 MG COMPRIMIDOS BUCODISPERSABLES EFG

Cómo usar RIZATRIPTAN FLAS CINFA 10 MG COMPRIMIDOS BUCODISPERSABLES EFG
Traducción generada por IA
Este contenido ha sido traducido automáticamente y se ofrece solo con fines informativos. No sustituye la consulta con un profesional sanitario.
Ver originalContenido del prospecto
Introducción
Prospecto: información para el usuario
Rizatriptán flas cinfa 10 mg comprimidos bucodispersables EFG
Lea todo el prospecto detenidamente antes de empezar a tomar este medicamento, porque contiene información importante para usted.
- Conserve este prospecto, ya que puede tener que volver a leerlo.
- Si tiene alguna duda, consulte a su médico o farmacéutico.
- Este medicamento se le ha recetado solamente a usted, y no debe dárselo a otras personas aunque tengan los mismos síntomas que usted, ya que puede perjudicarles.
- Si experimenta efectos adversos, consulte a su médico o farmacéutico, incluso si se trata de efectos adversos que no aparecen en este prospecto. Ver sección 4.
Contenido del prospecto
- Qué es rizatriptán flas cinfa y para qué se utiliza.
- Qué necesita saber antes de empezar a tomar rizatriptán flas cinfa.
- Cómo tomar rizatriptán flas cinfa.
- Posibles efectos adversos.
- Conservación de rizatriptán flas cinfa.
- Contenido del envase e información adicional.
1. Qué es rizatriptán flas y para qué se utiliza
Rizatriptán flas cinfa pertenece al grupo de medicamentos llamados agonistas selectivos de los receptores de serotonina 5-HT1B/1D.
Rizatriptán flas cinfa se usa para tratar los dolores de cabeza de las crisis de migraña en adultos. Tratamiento con rizatriptán flas cinfa:
Reduce la hinchazón de los vasos sanguíneos que rodean el cerebro. Esta hinchazón provoca el dolor de cabeza de una crisis de migraña.
2. Qué necesita saber antes de empezar a tomar rizatriptán flas cinfa
No tome rizatriptán flas cinfa
- si es alérgico al rizatriptán benzoato o a alguno de los demás componentes de este medicamento (incluidos en la sección 6).
- si tiene presión arterial alta moderadamente grave, grave, o leve que no está controlada con la medicación.
- si tiene o ha tenido alguna vez problemas de corazón, incluyendo infarto de miocardio o dolor en el pecho (angina) o ha experimentado signos relacionados con enfermedad de corazón.
- si tiene problemas graves de hígado o de riñón.
- si ha tenido un accidente cerebrovascular (ACV) o un accidente isquémico transitorio (AIT).
- si tiene problemas de obstrucción en sus arterias (enfermedad vascular periférica).
- si está tomando inhibidores de la monoaminooxidasa (MAO) como moclobemida, fenelcina, tranilcipromina o pargilina (medicamentos para la depresión), o linezolid (un antibiótico), o si han transcurrido menos de dos semanas desde que dejó de tomar un inhibidor de la MAO.
- si está tomado actualmente un medicamento de tipo ergotamínico, como ergotamina o dihidroergotamina para tratar su migraña o metisergida para prevenir una crisis de migraña.
- si está tomando cualquier otro medicamento de la misma clase, como sumatriptán, naratriptán, o zolmitriptán para tratar su migraña (ver más abajo Otros medicamentos y rizatriptán flas cinfa).
Si no está seguro de si alguno de los casos anteriores le afecta a usted, hable con su médico o farmacéutico antes de tomar rizatriptán flas cinfa.
Advertencias y precauciones
Consulte a su médico o farmacéutico antes de empezar a tomar rizatriptán flas cinfa.
Antes de tomar rizatriptán flas, informe a su médico o farmacéutico, si:
- tiene alguno de los siguientes factores de riesgo de enfermedad cardiaca: presión arterial alta, diabetes, es fumador o está usando sustitutivos de la nicotina, su familia tiene antecedentes de enfermedad cardiaca, es hombre y tiene más de 40 años o es una mujer postmenopáusica.
- tiene problemas de riñón o de hígado.
- tiene un problema determinado con la forma en la que su corazón late (bloqueo de rama izquierda).
- tiene o ha tenido alguna alergia.
- su dolor de cabeza está asociado con mareo, dificultad al caminar, falta de coordinación o debilidad en pierna y brazo.
- toma plantas medicinales que contienen hierba de San Juan.
- ha tenido reacciones alérgicas como hinchazón de la cara, labios, lengua y/o garganta que puede causar dificultad al respirar o tragar (angioedema).
- está tomando inhibidores selectivos de la recaptación de serotonina (ISRS) como sertralina, oxalato de escitalopram y fluoxetina o inhibidores de la recaptación de serotonina y noradrenalina (IRSN) como venlafaxina y duloxetina para la depresión.
- ha tenido síntomas pasajeros incluyendo dolor y opresión en el pecho.
Si toma rizatriptán flas con mucha frecuencia, esto puede producir que adquiera dolor de cabeza crónico. En tales casos debe contactar con su médico, ya que puede tener que dejar de tomar rizatriptán flas cinfa.
Informe a su médico o farmacéutico de todos sus síntomas. Su médico decidirá si tiene migraña. Sólo debe tomar rizatriptán flas cinfa para una crisis de migraña. Rizatriptán flas no debe usarse para tratar otros dolores de cabeza que puedan estar causados por otras enfermedades más graves.
Niños y adolescentes
No se recomienda el uso de rizatriptán flas cinfa comprimidos bucodispersables en niños y adolescentes menores de 18 años.
Otros medicamentos y rizatriptán flas cinfa
Informe a su médico o farmacéutico si está tomando, ha tomado recientemente o pudiera tener que tomar cualquier otro medicamento. Estos incluyen las plantas medicinales y aquellos medicamentos que tome normalmente para la migraña. Esto se debe a que rizatriptán flas cinfa puede afectar a la forma en la que funcionan algunos medicamentos. Otros medicamentos también pueden afectar a rizatriptán flas cinfa.
No tome rizatriptán flas cinfa:
- si ya está tomando un agonista 5-HT1B/1D (algunas veces denominados "triptanos"), como sumatriptán, naratriptán o zolmitriptán.
- si está tomando un inhibidor de la monoaminooxidasa (MAO), como moclobemida, fenelcina, tranilcipromina, linezolid o pargilina, o si han transcurrido menos de dos semanas desde que dejó de tomar un inhibidor de la MAO.
- si toma medicamentos de tipo ergotamínico, como ergotamina o dihidroergotamina para tratar su migraña.
- si toma metisergida para prevenir las crisis de migraña.
Los medicamentos arriba mencionados cuando se toman con rizatriptán flas cinfa pueden incrementar el riesgo de efectos adversos.
Después de tomar rizatriptán flas cinfa usted debe esperar al menos 6 horas antes de tomar medicamentos de tipo ergotamínico como ergotamina, dihidroergotamina o metisergida.
Después de tomar medicamentos de tipo ergotamínico usted debe esperar al menos 24 horas antes de tomar rizatriptán flas cinfa.
Pida a su médico instrucciones sobre cómo tomar rizatriptán flas cinfa e información sobre los riesgos
- si actualmente está tomando propranolol (ver sección 3 Cómo tomar rizatriptán flas cinfa).
- si actualmente está tomando inhibidores selectivos de la recaptación de serotonina (ISRS) como sertralina, oxalato de escitalopram y fluoxetina o inhibidores de la recaptación de serotonina y noradrenalina (IRSN) como venlafaxina y duloxetina para la depresión.
Toma de rizatriptán flas cinfa con alimentos y bebidas
Rizatriptán flas puede tardar más en hacer efecto si lo toma después de las comidas. Aunque es mejor tomarlo con el estómago vacío, usted puede tomarlo todavía aunque haya comido.
Embarazo y lactancia
Si está embarazada o en periodo de lactancia, cree que podría estar embarazada o tiene intención de quedarse embarazada, consulte a su médico o farmacéutico antes de utilizar este medicamento.
Los datos disponibles sobre la seguridad de rizatriptán cuando se usa durante los 3 primeros meses del embarazo no indican un aumento del riesgo de defectos de nacimiento. Se desconoce si rizatriptán flas es perjudicial para el feto cuando lo toma una mujer embarazada después de los 3 primeros meses del embarazo.
Si está en periodo de lactancia, puede posponer la lactancia durante 12 horas después del tratamiento para evitar la exposición del bebé.
Uso en pacientes mayores de 65 años
No hay estudios completos que evalúen la seguridad y la eficacia de rizatriptán flas en pacientes mayores de 65 años.
Conducción y uso de máquinas
Este medicamento al igual que la migraña puede causar somnolencia. Si nota estos efectos evite conducir o utilizar máquinas ya que puede ser peligroso.
Rizatriptán flas cinfa contiene lactosa
Este medicamento contiene lactosa. Si su médico le ha indicado que padece una intolerancia a ciertos azúcares, consulte con él antes de tomar este medicamento.
Rizatriptán flas cinfa contiene aspartamo
Este medicamento contiene 3,90 mg de aspartamo en cada comprimido.
El aspartamo contiene una fuente de fenilalanina que puede ser perjudicial en caso de padecer fenilcetonuria (FCN), una enfermedad genética rara en la que la fenilalanina se acumula debido a que el organismo no es capaz de eliminarla correctamente.
3. Cómo tomar rizatriptán flas cinfa
Rizatriptán flas cinfa se utiliza para tratar las crisis de migraña. Tome rizatriptán flas cinfa tan pronto como sea posible una vez comenzado su dolor de cabeza migrañoso. No lo utilice para prevenir una crisis.
Siga exactamente las instrucciones de administración de este medicamento indicadas por su médico o farmacéutico. En caso de duda, consulte de nuevo a su médico o farmacéutico.
La dosis recomendada es 10 mg.
Si actualmente está tomando propranolol o tiene problemas de riñón o de hígado, debe usar la dosis de 5 mg de rizatriptán. Debe esperar por lo menos 2 horas entre la toma de propranolol y la de rizatriptán hasta un máximo de 2 dosis en un periodo de 24 horas.
Si la migraña reaparece en 24 horas
En algunos pacientes, los síntomas de migraña pueden reaparecer en un periodo de 24 horas. Si su migraña reaparece, puede tomar una dosis adicional de rizatriptán flas cinfa. Debe esperar siempre por lo menos 2 horas entre las tomas.
Si después de 2 horas todavía tiene migraña
Si usted no responde a la primera dosis de rizatriptán flas cinfa durante una crisis, no debe tomar una segunda dosis de rizatriptán flas cinfa para el tratamiento de la misma crisis. Sin embargo, todavía es probable que responda a rizatriptán flas cinfa durante la siguiente crisis.
No tome más de 2 dosis de rizatriptán flas cinfa en un periodo de 24 horas (por ejemplo, no tome más de dos comprimidos de 10 mg en un periodo de 24 horas). Siempre debe esperar al menos 2 horas entre las tomas.
Si su estado empeora, busque atención médica.
Cómo administrar rizatriptán flas cinfa comprimidos bucodispersables
Rizatriptán flas cinfa (rizatriptán benzoato) está disponible en comprimidos bucodispersables de 10 mg que se disuelve en la boca.
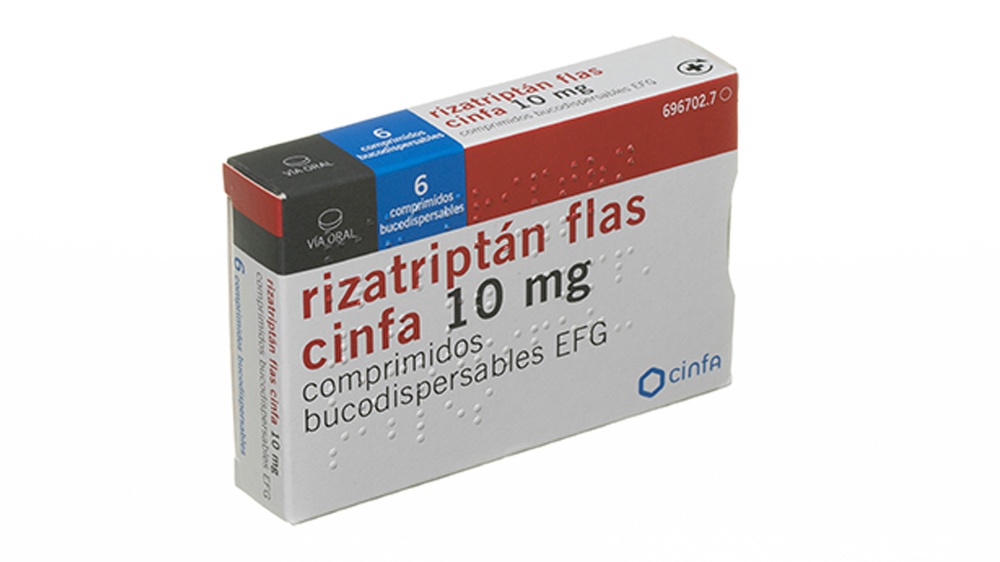
- No aplaste el comprimido bucodispersable
Para evitar que el comprimido bucodispersable se aplaste, no presionar el alveolo (Figura 1).
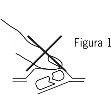
- Separe un alveolo
Cada blíster contiene seis alveolos, que están separados por perforaciones. Separe un alveolo siguiendo las líneas perforadas (Figura 2).
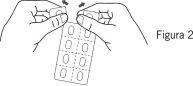
- Retire la lámina
Retire cuidadosamente la lámina, comenzando por la esquina donde se indica “despegar aluminio” (Figuras 3 y 4).
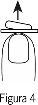
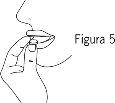
- Saque el comprimido bucodispersable
Saque el comprimido bucodispersable con las manos secas y póngalo en su lengua, donde se disolverá de forma que pueda ser tragado con la saliva (Figura 5).
El comprimido bucodispersable puede usarse en situaciones en las que no hay líquidos disponibles, o para evitar las náuseas y los vómitos que pueden acompañar la ingestión de comprimidos con líquidos.
Rizatriptán también está disponible en comprimidos para tomar con líquidos.
Si toma más rizatriptán flas cinfa del que debe
En caso de sobredosis o ingestión accidental consulte inmediatamente a su médico o farmacéutico o llame al Servicio de Información Toxicológica, teléfono 91 562 04 20, indicando el medicamento y la cantidad ingerida. Se recomienda llevar el envase y el prospecto del medicamento al profesional sanitario.
Los signos de sobredosis pueden incluir mareo, adormecimiento, vómitos, desfallecimiento y ritmo cardiaco lento.
Si tiene cualquier otra duda sobre el uso de este medicamento, pregunte a su médico o farmacéutico.
4. Posibles efectos adversos
Al igual que todos los medicamentos, este medicamento puede producir efectos adversos, aunque no todas las personas los sufran. Los siguientes efectos adversos pueden producirse con este medicamento.
En los estudios en adultos, los efectos adversos comunicados con más frecuencia fueron mareos, somnolencia y cansancio.
Frecuentes (pueden afectar hasta 1 de cada 10 pacientes)
- sensación de hormigueo (parestesia), dolor de cabeza, menor sensibilidad en la piel (hipoestesia), disminución de la agudeza mental, insomnio.
- ritmo cardiaco rápido o irregular (palpitación).
- rubor (enrojecimiento de la cara que dura un breve tiempo).
- molestias de garganta.
- malestar (nauseas), boca seca, vómitos, diarrea, indigestión (dispepsia).
- pesadez en partes del cuerpo, dolor de cuello, entumecimiento.
- dolor de abdomen o de pecho.
Poco frecuentes (pueden afectar hasta 1 de cada 100 pacientes)
- mal sabor en la boca.
- inestabilidad al andar (ataxia), mareos (vértigo), visión borrosa, temblor, desmayos (síncope).
- confusión, nerviosismo.
- presión arterial alta (hipertensión); sed, sofocos, sudoración.
- erupción cutánea, picor y erupción con bultos (urticaria); hinchazón de la cara, labios, lengua o garganta que puede causar dificultad para respirar o para tragar (angioedema), dificultad para respirar (disnea).
- sensación de rigidez en partes del cuerpo, debilidad muscular.
- cambios en el ritmo o la frecuencia del latido cardíaco (arritmia); alteraciones del electrocardiograma (una prueba que registra la actividad eléctrica de su corazón), ritmo cardiaco muy rápido (taquicardia).
- dolor facial; dolor muscular.
Raros (pueden afectar hasta 1 de cada 1.000 pacientes)
- ruidos al respirar.
- reacción alérgica (hipersensibilidad); reacción alérgica repentina y potencialmente mortal (anafilaxis).
- accidente cerebrovascular (esto ocurre generalmente en pacientes con factores de riesgo de enfermedad cardíaca o de vasos sanguíneos (hipertensión, diabetes, fumador, utilización de sustitutivos de la nicotina, historia familiar de enfermedad cardíaca o accidente cerebrovascular, hombre mayor de 40 años, mujer postmenopáusica y un problema determinado con la forma en la que su corazón late (bloqueo de rama izquierda))).
- latido cardíaco lento (bradicardia).
Frecuencia no conocida (no puede estimarse a partir de los datos disponibles)
- infarto de miocardio, espasmos de los vasos sanguíneos del corazón (estos ocurren generalmente en pacientes con factores de riesgo de enfermedad cardíaca o de vasos sanguíneos (hipertensión, diabetes, fumador, utilización de sustitutivos de la nicotina, historia familiar de enfermedad cardíaca o accidente cerebrovascular, hombre mayor de 40 años, mujer postmenopáusica y un problema determinado con la forma en la que su corazón late (bloqueo de rama izquierda).
- un síndrome llamado "síndrome serotoninérgico" que puede causar efectos adversos como coma, presión arterial inestable, fiebre extremadamente alta, falta de coordinación muscular, agitación y alucinaciones.
- descamación grave de la piel con o sin fiebre (necrólisis epidérmica tóxica).
- ataques (convulsiones/espasmos).
- contracción de los vasos sanguíneos de las extremidades incluyendo enfriamiento y adormecimiento de manos y pies.
- contracción de los vasos sanguíneos del colon (intestino grueso), lo cual puede causar dolor abdominal.
Informe inmediatamente a su médico si tiene síntomas de reacción alérgica, síndrome serotoninérgico, ataque al corazón o accidente cerebrovascular.
Además, informe a su médico si experimenta cualquier síntoma que indique una reacción alérgica (como erupción o picor) después de tomar rizatriptán flas.
Comunicación de efectos adversos
Si experimenta cualquier tipo de efecto adverso, consulte a su médico o farmacéutico, incluso si se trata de posibles efectos adversos que no aparecen en este prospecto. También puede comunicarlos directamente a través del Sistema Español de Farmacovigilancia de Medicamentos de Uso Humano: https://www.notificaram.es. Mediante la comunicación de efectos adversos usted puede contribuir a proporcionar más información sobre la seguridad de este medicamento.
5. Conservación de rizatriptán flas cinfa
Mantener este medicamento fuera de la vista y del alcance de los niños.
No conservar a temperatura superior a 30º C.
No utilice este medicamento después de la fecha de caducidad que aparece en el envase después de CAD. La fecha de caducidad es el último día del mes que se indica.
Los medicamentos no se deben tirar por los desagües ni a la basura. Deposite los envases y los medicamentos que no necesita en el Punto SIGRE de la farmacia. En caso de duda pregunte a su farmacéutico cómo deshacerse de los envases y de los medicamentos que no necesita. De esta forma ayudará a proteger el medio ambiente.
6. Contenido del envase e información adicional
Composición de rizatriptán flas cinfa
- El principio activo es rizatriptán. Cada comprimido bucodispersable contiene 10 mg de rizatriptán, equivalentes a 14,52 mg de rizatriptán benzoato.
- Los demás componentes son: lactosa monohidrato, celulosa microcristalina (E-460a), silicato cálcico, crospovidona, aspartamo (E-951), aroma de menta (contiene aroma natural y extracto de menta, maltodextrina derivada de maíz, almidón de maíz modificado, (E-1450)), sílice coloidal anhidra y estearato de magnesio.
Aspecto del producto y contenido del envase
Los comprimidos bucodispersables de 10 mg son de color blanco o casi blanco, con caras planas, redondos y con bordes biselados.
Rizatriptán flas cinfa 10 mg comprimidos se presenta en envases blíster de peel-off aluminio/aluminio de 2 o 6 comprimidos.
Titular de la autorización de comercialización y responsable de la fabricación
Titular de la autorización de comercialización
Laboratorios Cinfa, S.A.
Carretera Olaz-Chipi, 10. Polígono Industrial Areta
31620 Huarte (Navarra) - España
Responsable de la fabricación
Neuraxpharm Pharmaceuticals, S.L.
Avda. Barcelona, 69
08970 Sant Joan Despí
Barcelona – España
Fecha de la última revisión de este prospecto: Mayo 2024
La información detallada y actualizada de este medicamento está disponible en la página web de la Agencia Española de Medicamentos y Productos Sanitarios (AEMPS) http://www.aemps.gob.es/
Puede acceder a información detallada y actualizada sobre este medicamento escaneando con su teléfono móvil (smartphone) el código QR incluido en prospecto y cartonaje. También puede acceder a esta información en la siguiente dirección de internet: https://cima.aemps.es/cima/dochtml/p/77121/P_77121.html
Código QR a: https://cima.aemps.es/cima/dochtml/p/77121/P_77121.html
- País de registro
- Precio medio en farmacia8.8 EUR
- Principio activo
- Requiere recetaSí
- Fabricante
- Esta información es de carácter general y no sustituye la consulta con un profesional sanitario.
- Alternativas a RIZATRIPTAN FLAS CINFA 10 MG COMPRIMIDOS BUCODISPERSABLES EFGForma farmacéutica: COMPRIMIDO, 10 mgPrincipio activo: rizatriptanFabricante: Organon Salud S.L.Requiere recetaForma farmacéutica: COMPRIMIDO BUCODISPERSABLE/LIOTAB, 10 mgPrincipio activo: rizatriptanFabricante: Organon Salud S.L.Requiere recetaForma farmacéutica: COMPRIMIDO BUCODISPERSABLE/LIOTAB, 10 mg / comprimidoPrincipio activo: rizatriptanFabricante: Aurovitas Spain, S.A.U.Requiere receta
Médicos online para RIZATRIPTAN FLAS CINFA 10 MG COMPRIMIDOS BUCODISPERSABLES EFG
Comenta la dosis, los posibles efectos secundarios, interacciones, contraindicaciones o la revisión de receta de RIZATRIPTAN FLAS CINFA 10 MG COMPRIMIDOS BUCODISPERSABLES EFG, sujeto a valoración médica y a la normativa local.
Preguntas frecuentes















