
ALOCUTAN 50 MG/ML SOLUCION PARA PULVERIZACION CUTANEA

Cómo usar ALOCUTAN 50 MG/ML SOLUCION PARA PULVERIZACION CUTANEA
Traducción generada por IA
Este contenido ha sido traducido automáticamente y se ofrece solo con fines informativos. No sustituye la consulta con un profesional sanitario.
Ver originalContenido del prospecto
Introducción
Prospecto:información para el paciente
Alocutan50 mg/ml solución para pulverización cutánea
Minoxidil
Lea todo el prospecto detenidamente antes de empezar a usar este medicamento, porque contiene información importante para usted.
Siga exactamente las instrucciones de administración del medicamento contenidas en este prospecto o las indicadas por su médico o farmacéutico.
- Conserve este prospecto, ya que puede tener que volver a leerlo.
- Si necesita consejo o más información, consulte a su farmacéutico.
- Si experimenta efectos adversos, consulte a su médico o farmacéutico, incluso si se trata de efectos adversos que no aparecen en este prospecto. Ver sección 4.
- Debe consultar a un médico si empeora o si no mejora después de 4 meses.
Contenido del prospecto
- Qué es Alocutan 50 mg/ml y para qué se utiliza
- Qué necesita saber antes de empezar a usar Alocutan 50 mg/ml
- Cómo usar Alocutan 50 mg/ml
- Posibles efectos adversos
- Conservación de Alocutan 50 mg/ml
- Contenido del envase e información adicional
1. Qué es Alocutan 50 mg/ml y para qué se utiliza
Alocutan 50 mg / ml previene la pérdida de cabello hereditaria en hombres de entre 18 y 65 años y estimula el crecimiento del cabello. También promueve la regeneración de cabello nuevo en las zonas ya debilitadas o calvas y espesa el cabello existente.
Después de la aplicación, Alocutan 50 mg / ml penetra en el cuero cabelludo y llega a las raíces del cabello. Todavía no ha sido posible aclarar el mecanismo por el cual se estimula el crecimiento del cabello. Sin embargo, el principio activo de Alocutan 50 mg / ml puede detener la pérdida de cabello hereditaria, mediante:
- aumento el diámetro del tallo del cabello
- estimular el crecimiento y
- prolongación de las fases de crecimiento del cabello
Sin embargo, se necesitarán al menos cuatro meses antes de ver ningún resultado. Si las raíces del cabello todavía están presentes, el cabello fino y débil puede volver a crecer, haciéndose más largo, más denso y más oscuro con el tiempo. A qué velocidad comienza el crecimiento del cabello y cuántos cabellos vuelven a crecer depende del paciente. Las grandes calvas o pérdida de cabello presente durante más de 10 años son menos sensibles a Alocutan 50 mg / ml.
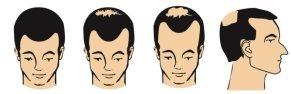
Las siguientes imagenes muestran las etapas de la pérdida de cabello de patrón masculino, para las cuales se ha podido demostrar la efectividad del minoxidil, la sustancia activa en Alocutan 50 mg / ml.
Niños y adolescentes menores de 18 años, pacientes de 65 años en adelante
Alocutan 50 mg / ml no debe utilizarse en estos grupos de pacientes, ya que no existen resultados de eficacia y seguridad de estudios disponibles en estos grupos de edad.
2. Qué necesita saber antes de empezar a usar Alocutan 50 mg/ml
No useAlocutan50 mg / ml
- si es alérgico al minoxidil o a cualquiera de los demás componentes de este medicamento (incluidos en la sección 6)
- si es mujer, ya que hay signos ocasionales de crecimiento del vello facial cosméticamente molesto y reversible durante el tratamiento
- si está usando otros medicamentos en el cuero cabelludo
- si tiene algún tipo de vendaje o apósito en el cuero cabelludo
- si sufre pérdida repentina o desigual de cabello
- si padece alguna afección que afecte su cuero cabelludo, incluida la psoriasis (afección cutánea inflamatoria con comezón), quemaduras solares, afeitado del cuero cabelludo o si la piel del cuero cabelludo está dañada por cicatrices o quemaduras, ya que no hay raíces capilares.
Advertencias y precauciones
Consulte a su médico o farmacéutico antes de usar Alocutan 50 mg / ml.
Las causas hormonales, las enfermedades subyacentes o la desnutrición se deben excluir. En estos casos, se necesita un tratamiento específico.
Aplique Alocutan 50 mg / ml solo en el cuero cabelludo sano normal. No debe usar Alocutan 50 mg / ml si no se conoce la causa de la pérdida de cabello o si la pérdida de cabello se debe a un parto o si el cuero cabelludo está enrojecido, inflamado o dolorido.
No hay experiencia clínica hasta la fecha con respecto a su efectividad para la pérdida de cabello en la región temporal (retroceso del cabello).
Si se producen efectos en los órganos internos (efectos sistémicos) o reacciones cutáneas graves, el tratamiento debe suspenderse.
Alocutan 50 mg / ml está destinado solo para uso externo sobre el cuero cabelludo. No aplicar Alocutan 50 mg / ml a otras partes del cuerpo.
No debe usar Alocutan 50 mg / ml
- si tiene algún signo de enfermedad cardiovascular o problemas de ritmo cardíaco
- si sufre de presión arterial alta
- si está usando medicamentos para tratar la presión arterial alta (agentes antihipertensivos).
Debe dejar Alocutan 50 mg / ml y consultar a un médico
- si tiene presión arterial baja o
- si aparecen uno o más de los siguientes síntomas: dolor en el pecho, latidos cardiacos más rápidos, debilidad o mareos, aumento repentino e inexplicable de peso, hinchazón de manos o pies, enrojecimiento persistente o irritación del cuero cabelludo o si aparecen otros nuevos síntomas no esperados (consulte la sección "Si usa más Alocutan 50 mg / ml del que debiera ").
El crecimiento de vello no deseado puede ser causado por la transferencia del producto a otras áreas distintas del cuero cabelludo.
Se ha informado de casos aislados de ligeros cambios en el color del cabello en pacientes con cabello muy rubio al usar otros productos para el cuidado del cabello al mismo tiempo, o después de nadar en agua muy clorada.
La ingestión accidental puede causar efectos secundarios graves en el sistema cardiovascular. Por lo tanto, debe mantener este medicamento fuera del alcance de los niños.
Se han notificado casos de crecimiento excesivo del vello corporal en lactantes tras el contacto de la piel con las zonas de aplicación de minoxidil en los pacientes (cuidadores) que usaban minoxidil tópico. El crecimiento del vello se normalizó en cuestión de meses, cuando los lactantes dejaron de estar expuestos a minoxidil. Se debe tener precaución para garantizar que los niños no entren en contacto con las zonas del cuerpo en las que se haya aplicado minoxidil por vía tópica.
Consulte al médico si observa un crecimiento excesivo del vello en el cuerpo de su hijo durante el período en que utilice productos tópicos con minoxidil.
Evite respirar la neblina de pulverización.
Niños y adolescentes
No administre este medicamento a niños y adolescentes menores de 18 años, ya que no se ha establecido la seguridad ni la eficacia en este grupo de edad.
Otros medicamentos yAlocutan50 mg/ml
Informe a su médico o farmacéutico si está utilizando / tomando, ha tomado / utilizado recientemente o podría tomar / usar cualquier otro medicamento.
Hasta la fecha, no hay información disponible sobre las interacciones entre Alocutan 50 mg / ml y otros principios activos. Aunque no está clínicamente probado, existe una posibilidad teórica de que la absorción de la sustancia activa en Alocutan 50 mg / ml (minoxidil) en el cuerpo pueda empeorar la hipotensión ortostática (una disminución de la presión arterial al levantarse desde una posición acostada) en pacientes que también están tomando vasodilatadores periféricos (ciertos medicamentos para la presión arterial alta que ensanchan los vasos sanguíneos).
Alocutan 50 mg / ml no debe utilizarse junto con otros productos dermatológicos (preparados para uso externo que contengan sustancias activas como corticosteroides, retinoides o ditranol) o con principios activos que aumenten la absorción de la sustancia activa a través de la piel (absorción cutánea) .
Embarazo y lactancia
Alocutan 50 mg / ml está indicado solo para pacientes varones.
La experiencia es limitada con la aplicación de Alocutan 50 mg / ml durante el embarazo. Minoxidil absorbido por el cuerpo puede pasar a la leche materna. Por lo tanto, Alocutan 50 mg / ml no debe ser utilizado por mujeres embarazadas y mujeres en período de lactancia.
Conducción y uso de máquinas
Este producto puede causar mareos o hipotensión (ver sección 4). Si se ve afectado, no debe conducir ni utilizar máquinas.
Alocutan50 mg/ml contiene propilenglicol (E 1520) y etanol 96% (alcohol)
Este medicamento contiene 509 mg de propilenglicol (E 1520) en cada ml de solución.
Este medicamento contiene 248 mg de alcohol (etanol) en cada ml de solución.
Puede causar sensación de ardor en piel lesionada
El etanol puede causar picor e irritación en los ojos. En caso de contacto accidental con áreas sensibles (ojos, abrasiones de la piel, membranas mucosas), debe enjuagarlas con abundante agua.
Si aplica Alocutan 50 mg / ml repetidamente en el cabello en lugar de en el cuero cabelludo, esto podría aumentar la sequedad y /o la rigidez del cabello debido al contenido de etanol y propilenglicol en Alocutan 50 mg / ml.
3. Cómo usar Alocutan 50 mg/ml
Use siempre este medicamento exactamente como se describe en este prospecto o como le haya indicado su médico o farmacéutico. Consulte con su médico o farmacéutico si no está seguro.
Alocutan 50 mg / ml es para uso externo en el cuero cabelludo seco. Utilice Alocutan 50 mg / ml solo en el cuero cabelludo sano y sin daños y siga las instrucciones de uso a continuación exactamente en todo momento. No aplique Alocutan 50 mg / ml en áreas del cuerpo que no sean el cuero cabelludo.
La dosis recomendada es:
Aplique 1 ml de Alocutan 50 mg / ml dos veces al día (mañana y tarde) en las zonas afectadas del cuero cabelludo.
No debe excederse la cantidad diaria aplicada, es decir, solución de 2 x 1 ml, independientemente del tamaño del área afectada de la piel.
Uso en niños y adolescentes
Alocutan 50 mg / ml no debe usarse en niños y adolescentes menores de 18 años, ya que no hay resultados disponibles de seguridad y eficacia de estudios controlados para estos grupos de edad.
Forma de administración
Uso cutáneo (cuero cabelludo).
Cada envase de Alocutan 50 mg / ml contiene 2 aplicadores pulverizadores diferentes:
- aplicador premontado para aplicaciones de grandes superficies
- aplicador separado con punta extendida para áreas más pequeñas
Ambos aplicadores se pueden intercambiar separando el aplicador y reemplazándolo por el otro.
Para una dosis de 1 ml, se requieren 6 pulverizaciones.
Instrucciones de uso / aplicación:
La solución se rocía directamente sobre el cuero cabelludo dentro del área de pérdida de cabello. Para esto, presione hacia abajo la bomba seis veces. Después de cada acción de bombeo, extienda el líquido sobre el área afectada con las yemas de los dedos. Al mismo tiempo, evite inhalar la niebla de la pulverización.
Qué más debe tener en cuenta durante el uso
Las manos deben lavarse cuidadosamente después de aplicar Alocutan 50 mg / ml, para evitar el contacto accidental con las mucosas y los ojos.
Después de aplicar Alocutan 50 mg / ml, puede peinar su cabello de forma normal. Sin embargo, no debe humedecer el cuero cabelludo durante aproximadamente 4 horas. Esto evitará que se elimine Alocutan 50 mg / ml
Duración del tratamiento
El inicio y el grado del crecimiento del cabello son diferentes en cada paciente.
En general, se requiere un tratamiento dos veces al día durante 2 a 4 meses antes de que se observe un efecto. Para mantener el efecto, se recomienda continuar la aplicación dos veces al día sin interrupción.
No obtendrá mejores resultados aplicando Alocutan 50 mg / ml en cantidades mayores o con mayor frecuencia. Con respecto a un posible efecto terapéutico, hay suficiente experiencia clínica para un período de tratamiento de hasta un año.
Si no se obtiene la respuesta terapéutica deseada dentro de los 4 meses, se debe interrumpir el tratamiento.
Información sobre el aumento de la pérdida de cabello
En el tratamiento de los folículos pilosos con el principio activo minoxidil, la fase de reposo del ciclo del pelo (fase telógena) se acorta y la fase de crecimiento (fase anágena) se alcanza más rápidamente. Esto estimula el crecimiento de cabello nuevo, que expulsa los cabellos "viejos", ya no activos, fuera del cuero cabelludo. Esto da la impresión inicial de una mayor pérdida de cabello. En algunos pacientes, esta reacción se ha observado de dos a seis semanas después del inicio del tratamiento con el principio activo minoxidil. Sin embargo, no hay necesidad de alarmarse, ya que esta reacción se acompaña de un aumento del crecimiento del cabello. El efecto desaparece en unas pocas semanas y puede interpretarse como una primera señal del efecto del minoxidil.
Comunique a su médico o farmacéutico si tiene la impresión de que el efecto de Alocutan 50 mg / ml es demasiado fuerte o demasiado débil
Si usa más Alocutan50 mg / ml del que debe
La aplicación de Alocutan 50 mg / ml a una dosis superior a la recomendada y en superficies corporales relativamente grandes o áreas distintas del cuero cabelludo puede conducir posiblemente a una mayor absorción de minoxidil en el cuerpo. Hasta la fecha, no se conocen casos en los que el uso externo de la solución de minoxidil haya provocado síntomas de intoxicación.
Después de la ingestión accidental, la concentración del componente activo minoxidil en Alocutan 50 mg / ml puede producir efectos en los órganos internos que corresponden a los efectos que resultan cuando se ingiere la sustancia activa, por ejemplo, en un comprimido. Esto puede causar los siguientes efectos secundarios: latido del corazón más rápido, disminución de la presión arterial, acumulación de líquido y posterior aumento repentino de peso, mareos.
En caso de ingestión accidental o si se presentan signos de sobredosis, informe a su médico de inmediato, para que él o ella pueda decidir qué hacer a continuación. Por favor, tenga a mano el propsecto del medicamento para que el médico pueda informarse sobre qué sustancia activa se ha tomado.
Si olvidó usar Alocutan50 mg / ml
No aplique una dosis doble para compensar una dosis olvidada; continúe su tratamiento a la dosis recomendada. Recuperar la dosis omitida no traerá ningún beneficio y podría provocar efectos indeseables.
Si interrumpe el tratamiento con Alocutan50 mg / ml
Es necesario continuar el tratamiento para mejorar y mantener el crecimiento del cabello. De lo contrario, la pérdida de cabello volverá a ocurrir.
Si el tratamiento se interrumpe dentro de 3 a 4 meses, los efectos corresponden a los que se hubieran logrado sin tratamiento con Alocutan 50 mg / ml.
Si tiene más preguntas sobre el uso de este medicamento, consulte a su médico o farmacéutico.
4. Posibles efectos adversos
Al igual que todos los medicamentos, este medicamento puede producir efectos adversos, aunque no todas las personas los sufran.
Póngase en contacto con un médico de inmediato si nota alguno de los siguientes síntomas: es posible que necesite tratamiento médico urgente.
- Hinchazón de la cara, los labios o la garganta que dificulta la ingestión o la respiración. Esto podría ser un signo de una reacción alérgica grave (frecuencia desconocida, no puede estimarse a partir de los datos disponibles)
- Enrojecimiento generalizado de la piel (frecuencia desconocida, no puede estimarse a partir de los datos disponibles)
- Picor generalizado (frecuencia desconocida, no puede estimarse a partir de los datos disponibles)
- Opresión en la garganta (frecuencia desconocida, no puede estimarse a partir de los datos disponibles)
Muy frecuentes(puede afectar a más de 1 de cada 10 personas):
- Dolor de cabeza
Frecuentes(puede afectar hasta 1 de cada 10 personas):
- Picor
- Aumento del crecimiento del cabello más allá del cuero cabelludo (incluido el crecimiento del vello facial en las mujeres), reacción inflamatoria de la piel (que incluye erupción acneiforme, erupción cutánea)
- Dificultad para respirar, falta de aliento
- Brazos y piernas hinchados
- Aumento de peso
- Alta presión sanguínea
- Irritación del cuero cabelludo como escozor, ardor, sequedad, comezón, descamación, foliculitis
Poco frecuentes(pueden afectar hasta 1 de cada 100 personas):
- Mareos, náuseas
Desconocido(la frecuencia no puede estimarse a partir de los datos disponibles):
- Dermatitis de contacto (inflamación de la piel relacionada con la alergia)
- Estado del ánimo deprimido
- Irritación de ojo
- Latido del corazón más rápido, palpitaciones, presión arterial baja
- Vómitos
- Síntomas en el sitio de administración que también pueden afectar los oídos y la cara, como picor, irritación de la piel, dolor, enrojecimiento, hinchazón, piel seca y erupción inflamatoria hasta posible descamación de la piel, inflamación de la piel (dermatitis), ampollas, sangrado y ulceración
- Pérdida temporal de cabello, cambios en el color del cabello, estructura alterada del cabello
- Dolor en el pecho
Comunicación de efectos adversos
Si experimenta cualquier tipo de efecto adverso, consulte a su médico o farmacéutico, incluso si se trata de efectos adversos que no aparecen en este prospecto. También puede comunicarlos directamente a través del sistema nacional de notificación incluido en el Sistema Español de Farmacovigilancia de Medicamentos de Uso Humano: www.notificaram.es. Mediante la comunicación de efectos adversos puede contribuir a proporcionar más información sobre la seguridad de este medicamento.
5. Conservación de Alocutan 50 mg/ml
Mantenga este medicamento fuera de la vista y del alcance de los niños.
No use este medicamento después de la fecha de caducidad que figura en el envase y la botella después de "EXP". La fecha de caducidad se refiere al último día de ese mes.
No congelar.
Contiene etanol que es inflamable. Almacenar lejos de fuentes de calor o llamas.
Nota sobre la caducidad una vez abierto
Después de abrir el frasco, use la solución dentro de las 6 semanas.
Los medicamentos no se deben tirar por los desagües ni a la basura. Deposite los envases y los medicamentos que no necesita en el Punto SIGRE de la farmacia. En caso de duda pregunte a su farmacéutico cómo deshacerse de los envases y de los medicamentos que no necesita. De esta forma ayudará a proteger el medio ambiente.
6. Contenido del envase e información adicional
Composición deAlocutan50 mg/ml
El principio activo es minoxidil.
1 ml de solución contiene 50 mg de minoxidil.
Los demás componentes son: etanol al 96% (v / v), propilenglicol (E1520), agua purificada.
Aspecto deAlocutan 50 mg / ml y contenido del envase
Alocutan 50 mg / ml es una solución clara, incolora a amarilla pálida y está disponible en envases de 60 ml y 3 x 60 ml de solución cutánea en frascos de HDPE blancos.
Cada envase contiene 2 aplicadores diferentes, un aplicador de pulverizador de bomba premontado y un aplicador con punta extendida.
Titular de la autorización de comercialización y responsable de la fabricación
Titular de la autorización de comercialización
Mibe Pharma España S.L.U.
C/Amaltea 9, 4ª planta, letra B,
28045, Madrid
España
Responsable de la fabricación
Mibe GmbH Arzneimittel
Münchener Strasse, 15
06796 Brehna
Alemania
Este medicamento está autorizado en los Estados miembros del EEE con los siguientes nombres:
Austria: Minoxidil Dermapharm 50 mg/ml Spray zur Anwendung auf der Haut, Lösung
Italia: Phalanx 50 mg/ml spray cutaneo, soluzione
España: Alocutan 50 mg/ml solución para pulverización cutánea
Fecha de la última revisión de este prospecto:Octubre 2024
La información detallada de este medicamento está disponible en la página web de la Agencia Española de Medicamentos y Productos Sanitarios (AEMPS) (http://www.aemps.gob.es/).
- País de registro
- Principio activo
- Requiere recetaNo
- Fabricante
- Esta información es de carácter general y no sustituye la consulta con un profesional sanitario.
- Alternativas a ALOCUTAN 50 MG/ML SOLUCION PARA PULVERIZACION CUTANEAForma farmacéutica: LIQUIDO USO TOPICO, 5%Principio activo: MinoxidilFabricante: Laboratorios Vinas S.A.No requiere recetaForma farmacéutica: LIQUIDO USO TOPICO, 50 mg/mlPrincipio activo: MinoxidilFabricante: Laboratorios Serra Pamies S.A.No requiere recetaForma farmacéutica: LIQUIDO USO TOPICO, 2000 mgPrincipio activo: MinoxidilFabricante: Laboratorios Galderma S.A.No requiere receta
Médicos online para ALOCUTAN 50 MG/ML SOLUCION PARA PULVERIZACION CUTANEA
Comenta la dosis, los posibles efectos secundarios, interacciones, contraindicaciones o la revisión de receta de ALOCUTAN 50 MG/ML SOLUCION PARA PULVERIZACION CUTANEA, sujeto a valoración médica y a la normativa local.
Preguntas frecuentes








