
How to use CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETS
Translated with AI
This page provides general information and does not replace a doctor’s consultation. Always consult a doctor before taking any medication. Seek urgent medical care if symptoms are severe.
Show originalContents of the leaflet
Introduction
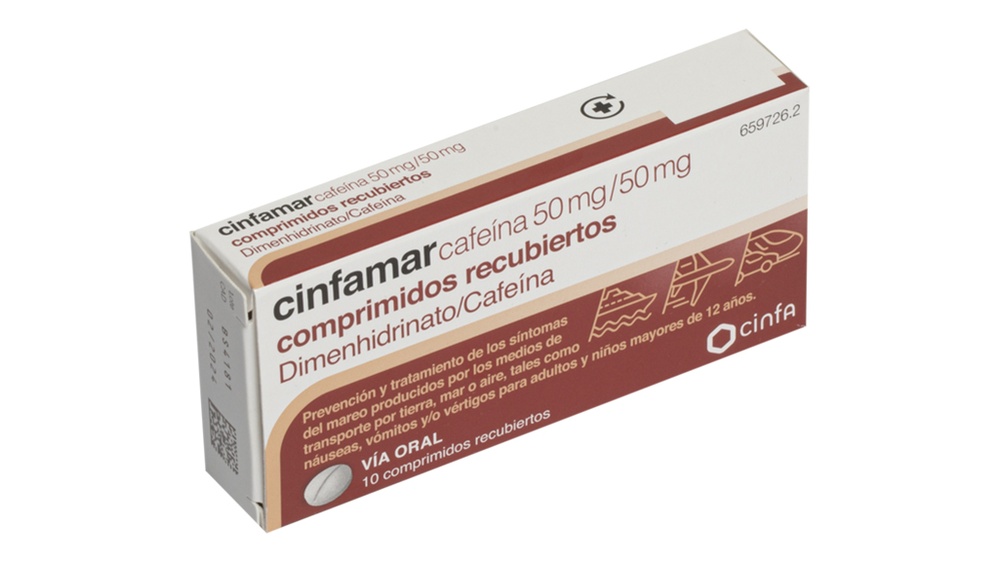
Leaflet: information for the user
cinfamar caffeine 50mg/50mg coated tablets
dimenhydrinate/caffeine
Read the entire leaflet carefully before starting to take this medicine, as it contains important information for you.
- Follow the administration instructions for the medicine contained in this leaflet or as indicated by your doctor or pharmacist.
- Keep this leaflet, as you may need to read it again.
- If you need advice or more information, consult your pharmacist.
- If you experience side effects, consult your doctor or pharmacist, even if they are not listed in this leaflet. See section 4.
- You should consult a doctor if it worsens or does not improve after 7 days.
Contents of the leaflet
- What is cinfamar caffeine and what is it used for
- What you need to know before taking cinfamar caffeine
- How to take cinfamar caffeine
- Possible side effects
- Storage of cinfamar caffeine
- Package contents and additional information
1. What is cinfamar caffeine and what is it used for
cinfamar caffeine is a medicine that contains two active substances: dimenhydrinate and caffeine. Dimenhydrinate is an active ingredient that acts against motion sickness.
Caffeine has a stimulating effect on the nervous system, so it counteracts the drowsiness effect that dimenhydrinate can cause.
cinfamar caffeine is a medicine that is indicated for the prevention and treatment of symptoms of motion sickness caused by land, sea, or air travel, such as nausea, vomiting, and/or vertigo for adults and children over 12 years old.
You should consult a doctor if it worsens or does not improve after 7 days.
2. What you need to know before taking cinfamar caffeine
Do not take cinfamar caffeine
- If you are allergic to dimenhydrinate, diphenhydramine, caffeine (or its derivatives, such as aminophylline, theophylline, etc.) or any of the other components of this medicine (listed in section 6).
- If you have porphyria (a rare, usually hereditary disorder in which a large amount of porphyrin is eliminated in urine and feces).
- If you have asthma attacks.
- If you have mental disorders that cause nervous excitement or convulsive crises, with or without loss of consciousness (epilepsy).
- If you have anxiety or insomnia (difficulty sleeping).
- If you have severe liver or heart disorders.
- If you have stomach or duodenal ulcers.
- If you have high blood pressure.
Warnings and precautions
Consult your doctor or pharmacist before starting to take cinfamar caffeine.
- If you are allergic to any allergy medicine, you may also be allergic to dimenhydrinate.
- If you have asthma or severe respiratory diseases (chronic obstructive pulmonary disease, emphysema, chronic bronchitis), as it can cause thickening of secretions and alter expectoration.
- If you have a disease in which there is an abnormal increase in thyroid gland activity (hyperthyroidism), increased intraocular pressure (glaucoma), prostate enlargement (prostatic hypertrophy), or any disease that causes obstruction of the urinary or gastrointestinal tract.
- If you are over 65 years old, dry mouth, urinary retention, nausea, sedation, confusion, and low blood pressure (hypotension) may occur.
- If you are using any medicine that produces ear toxicity, which manifests with symptoms such as ringing in the ears, dizziness, or vertigo, as this medicine may mask the symptoms of those toxic effects.
- If you are diabetic, you should be aware that caffeine can raise blood sugar levels.
- If you have any liver, kidney, stomach, or duodenal ulcer disease, or stomach inflammation (gastritis), you should consult your doctor before taking this medicine.
- You should consult your doctor before taking this medicine if you have or have had heart diseases (cardiac arrhythmia, myocardial ischemia, etc.), especially when engaging in physical exercise or being in high-altitude areas.
- If you are sensitive to caffeine or other xanthines, such as theophylline, aminophylline, or others, you should consult your doctor before taking this medicine.
- If you suspect you may have appendicitis symptoms such as nausea, vomiting, or abdominal cramps, it is recommended to see a doctor to rule out the presence or absence of appendicitis, as dimenhydrinate can make it difficult to diagnose this disease.
- It is recommended to avoid exposure to very high temperatures and follow adequate hygiene and dietary measures, such as proper ventilation and hydration.
- Avoid exposure to the sun (even on cloudy days) and UVA lamps (ultraviolet rays) while taking this medicine.
- The simultaneous use of tobacco with the caffeine contained in this medicine can increase the degradation of caffeine.
Children
Children under 12 years old should not take this medicine.
Taking cinfamar caffeine with other medicines
Tell your doctor or pharmacist if you are taking, have recently taken, or may need to take any other medicine.
In particular, if you are using any of the following medicines, it may be necessary to modify the dose of cinfamar caffeine or not take it:
Medicines that primarily interfere with dimenhydrinate:
- Anesthetics and other substances with a central nervous system depressant effect.
- Antibiotics of the aminoglycoside group.
- Antidepressants, such as monoamine oxidase inhibitors (MAOIs).
- Antiparkinsonian agents.
- Neuroleptics (used to calm agitation and neuromuscular hyperactivity).
- Ototoxic medicines (which can affect the ear) (see Warnings and Precautions section).
- Medicines that can cause photosensitivity (skin reactions due to sensitivity to sunlight).
Medicines that primarily interfere with caffeine:
- Oral contraceptives.
- Anti-infectives of the quinolone group (e.g., pipemidic acid, ciprofloxacin), linezolid, and erythromycin.
- Barbiturates (used as anticonvulsants).
- Adrenergic bronchodilators and theophylline (used to combat bronchial asthma).
- Calcium.
- Cimetidine (used for the treatment of excessive gastric secretion and stomach ulcers).
- Disulfiram (used in alcohol withdrawal treatments).
- Phenytoin (used for the treatment of epilepsy).
- Iron (used for the treatment of anemia. Caffeine decreases iron absorption, so it should be taken at least 2 hours apart).
- Lithium (antidepressant).
- Mexiletine: used for heart rhythm disorders (cardiac arrhythmias).
- Procarbazine (anticancer).
- Selegiline (used for the treatment of Parkinson's disease).
- Thyroxine (used for the treatment of thyroid diseases).
Interference with diagnostic tests
If you are going to have any analytical tests (blood tests, urine tests, etc.), tell your doctor that you are taking this medicine, as it may alter the results of these tests.
If you are going to have any allergy tests, including skin tests, it is recommended to stop treatment 72 hours before starting the test, to avoid altering the results.
Additionally, diabetic patients should be aware that caffeine can increase blood glucose levels.
Taking cinfamar caffeine with food, drinks, and alcohol
It is not recommended to consume alcohol while taking this medicine.
Limit the consumption of beverages that contain caffeine (coffee, tea, chocolate, and cola drinks) while taking this medicine.
Pregnancy and breastfeeding
If you are pregnant or breastfeeding, think you may be pregnant, or plan to become pregnant, consult your doctor or pharmacist before using this medicine.
Breastfeeding
Women who are breastfeeding should not take this medicine without first consulting their doctor or pharmacist, as this medicine passes into breast milk.
Driving and using machines
While being treated with this medicine, do not drive or operate hazardous machinery, as this medicine can cause drowsiness or decreased reaction capacity at the recommended doses.
cinfamar caffeine contains sodium
This medicine contains less than 23 mg of sodium (1 mmol) per tablet; it is essentially "sodium-free".
3. How to take cinfamar caffeine
Follow the administration instructions for the medicine contained in this leaflet or as indicated by your doctor or pharmacist. In case of doubt, ask your doctor or pharmacist.
The recommended dose is:
Adults and children over 12 years old
1 or 2 tablets per dose. If necessary, repeat the dose every 4-6 hours.
Do not take more than 8 tablets per day (corresponding to 400 mg of dimenhydrinate and 400 mg of caffeine).
Patients with liver disease
They should consult their doctor before taking this medicine, as it may be necessary to adjust the dose.
Use in children
Children under 12 years old should not take this medicine.
cinfamar caffeine is administered orally.
The first dose should be taken at least 30 minutes before starting the trip (preferably 1 or 2 hours before), leaving at least 4 hours between one dose and the next. It is recommended to take the tablets with food, water, or milk to minimize gastric irritation.
The last dose should not be taken in the 6 hours prior to bedtime, to avoid possible insomnia, especially in patients with difficulty sleeping.
If it worsens or if the symptoms persist for more than 7 days, you should consult your doctor.
If you take more cinfamar caffeine than you should
In case of overdose or accidental ingestion, consult your doctor or pharmacist immediately or call the Toxicology Information Service, phone 91 562 04 20, indicating the medicine and the amount taken.
The symptoms of overdose include mainly: dilated pupils, red face, excitement, hallucinations, confusion, stomach and intestinal irritation with nausea, vomiting, and diarrhea, movement disorders, convulsions, deep loss of consciousness (coma), sudden decrease in respiratory and cardiac functions (cardiorespiratory collapse), and death. The symptoms may take more than 2 hours to appear after the overdose.
If you have any other doubts about the use of this medicine, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everyone gets them.
Side effects usually occur at the beginning of treatment.
During the use of dimenhydrinate, the following adverse reactions have been observed, whose frequency could not be established with precision:
- Nausea, vomiting, constipation, diarrhea, stomach pain.
- Lack of appetite and dry mouth.
- Sleepiness and sedation (drowsiness).
- Headache, dizziness, and vertigo.
- Increased viscosity of mucus in the bronchi, making breathing difficult.
- Urinary retention and impotence.
- Glaucoma (increased intraocular pressure).
- Pupil dilation, blurred vision, or double vision.
- Allergic reactions on the skin and sensitivity to sunlight after intense exposure, which can cause hives, itching, and skin redness.
- Decrease in the blood of the number of red blood cells, white blood cells, leukocytes, and platelets.
- Porphyria (a rare, usually hereditary disorder in which a large amount of porphyrin is eliminated in feces and urine).
- Hypotension (decreased blood pressure).
- Tachycardia, palpitations, and/or cardiac arrhythmias.
- Hyperexcitability may occur, especially in children, with symptoms such as insomnia, nervousness, confusion, tremors, irritability, euphoria, delirium, palpitations, and even convulsions.
Additionally, due to caffeine, the following may appear:
- Excitement, agitation, and/or difficulty sleeping.
- Ringing in the ears.
- Disorientation.
- Rapid breathing.
- Hot flashes.
- Abundant urine elimination.
- Hyperglycemia or hypoglycemia (increased or decreased glucose in the blood).
Reporting side effects
If you experience any type of side effect, consult your doctor or pharmacist, even if it is a possible side effect that is not listed in this leaflet. You can also report them directly through the Spanish Medicines and Health Products Agency (AEMPS) https://www.notificaram.es. By reporting side effects, you can contribute to providing more information on the safety of this medicine.
5. Storage of cinfamar caffeine
Keep this medicine out of the sight and reach of children.
This medicine does not require special storage conditions.
Do not use this medicine after the expiration date shown on the packaging, after EXP. The expiration date is the last day of the month indicated.
Medicines should not be disposed of through wastewater or household waste. Deposit the packaging and medicines that are no longer needed in the pharmacy's SIGRE point. If in doubt, ask your pharmacist how to dispose of the packaging and medicines that are no longer needed. This will help protect the environment.
6. Package contents and additional information
Composition of cinfamar caffeine
- Each tablet contains 50 milligrams of dimenhydrinate and 50 milligrams of caffeine as active ingredients.
- The other components are:
- Core: Cornstarch, anhydrous colloidal silica, microcrystalline cellulose, calcium phosphate dihydrate, magnesium stearate, sodium carboxymethyl starch (potato) (type A), povidone, talc.
- Coating: Macrogol 6000, hypromellose 606, hypromellose 615.
Appearance of the product and package contents
cinfamar caffeine are white, cylindrical, biconvex, and scored on one side coated tablets.
They come in packages containing 4 or 10 coated tablets.
Marketing authorization holder and manufacturer
Laboratorios Cinfa, S.A.
Carretera Olaz-Chipi, 10. Polígono Industrial Areta
31620 Huarte (Navarra) – Spain
Date of the last revision of this leaflet:December 2020
Detailed and updated information on this medicine is available on the website of the Spanish Agency for Medicines and Health Products (AEMPS) http://www.aemps.gob.es/
-

- Country of registration
- Dosage formTABLET, 50/50 mg/mg
- ATC codeR06AA52
- Active substance
- Prescription requiredNo
- Manufacturer
- This information is for reference only and does not constitute medical advice. Always consult a doctor before taking any medication. Oladoctor is not responsible for medical decisions based on this content.
- Alternatives to CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETSDosage form: TABLET, 50/15/50 mg/mg/mgActive substance: diphenhydramine, combinationsManufacturer: Uriach Consumer Healthcare S.L.Prescription not requiredDosage form: CHEWING GUM, 20 mg dimenhydrinateActive substance: diphenhydramineManufacturer: Uriach Consumer Healthcare S.L.Prescription not requiredDosage form: TABLET, 50 mgActive substance: diphenhydramineManufacturer: Uriach Consumer Healthcare S.L.Prescription not required
Online doctors for CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETS
Discuss questions about CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETS, including use, safety considerations and prescription review, subject to medical assessment and local regulations.
Get CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETS Prescription Online
Fill in a 2-min form
Tell us your symptoms, history, and what medicine you're requesting.
Pick a doctor or let us assign
Choose a specialist or we'll match you with the next available doctor.
Doctor reviews your case
Usually within 30 minutes. They may ask follow-up questions via chat.
Collect from any pharmacy
Electronic prescription sent to your email — valid across Spain.
Frequently Asked Questions
CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETS does not require a prescription in Spain. You can check with a doctor online whether this medicine may be appropriate for your situation.
The active ingredient in CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETS is diphenhydramine, combinations. This information helps identify medicines with the same composition but different brand names.
CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETS is manufactured by Laboratorios Cinfa S.A.. Pharmacy brands and packaging may differ depending on the distributor.
Doctors such as Family doctors, Psychiatrists, Dermatologists, Cardiologists, Endocrinologists, Gastroenterologists, Pulmonologists, Nephrologists, Rheumatologists, Hematologists, Infectious disease physicians, Allergists, Geriatricians, Paediatricians, Oncologists may assess whether CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETS is appropriate, depending on your situation and local regulations. You can book an online consultation to discuss your symptoms and possible next steps.
Spain has a well-developed healthcare infrastructure across major cities such as Madrid, Barcelona, Valencia, and Seville. Pharmacies are widely available and operate under strict regulations, ensuring access to prescribed medications.
You can buy CINFAMAR CAFFEINE 50 mg/50 mg FILM-COATED TABLETS in Madrid, Barcelona, Seville, or Valencia at any local pharmacy with a valid prescription.
To obtain a prescription, you can use Oladoctor:
Other medicines with the same active substance (diphenhydramine, combinations) include BIODRAMINA CAFFEINE COATED TABLETS, BIODRAMINA 20 mg CHEWABLE MEDICATED LOZENGES, BIODRAMINA 50 mg TABLETS. These may have different brand names or formulations but contain the same therapeutic ingredient. Always consult a doctor before switching or starting a new medicine.














