How to use RABIPUR Powder and Solvent for Injectable Solution in Pre-filled Syringe
Translated with AI
This page provides general information and does not replace a doctor’s consultation. Always consult a doctor before taking any medication. Seek urgent medical care if symptoms are severe.
Show originalContents of the leaflet
Introduction
Package Leaflet: Information for the User
Rabipur
Powder and solvent for solution for injection in a pre-filled syringe
rabies virus (inactivated, Flury LEP strain)
Read all of this leaflet carefully before you or your child receive Rabipur, because it contains important information for you.
- Keep this leaflet, you may need to read it again.
- If you have any further questions, ask your doctor, pharmacist, or nurse.
- This vaccine has been prescribed for you or your child. Do not pass it on to others.
- If you experience any side effects, talk to your doctor, pharmacist, or nurse, even if they are not listed in this leaflet. See section 4.
Contents of the package leaflet:
- What is Rabipur and what is it used for
- What you need to know before you or your child receive Rabipur
- How to use Rabipur
- Possible side effects
- Storage of Rabipur
- Contents of the pack and other information
1. What is Rabipur and what is it used for
What is Rabipur
Rabipur is a vaccine that contains the rabies virus that has been inactivated. After administration of the vaccine, the immune system (the body's natural defense system) forms antibodies against the rabies virus. These antibodies protect you from infections or diseases caused by the rabies virus. None of the vaccine components can cause rabies.
What Rabipur is used for
Rabipur can be used in people of all ages.
Rabipur can be used to prevent rabies:
- before a possible risk of exposure to the rabies virus (pre-exposure prophylaxis)
or
- after a suspected or confirmed exposure to the rabies virus (post-exposure prophylaxis).
Rabies is an infection that can be transmitted when an infected animal bites, scratches, or even licks a person, especially when the skin is not intact. Even contact with animal traps that have been licked or bitten by infected animals can cause infections in humans.
2. What you need to know before you or your child receive Rabipur
You or your child should not receive Rabipur before a possible risk of exposure to the rabies virus if:
- You have a history of severe allergic reactions to the active substance or to any of the other components of the vaccine (listed in section 6).
- You are suffering from an acute illness that requires treatment.
Due to the severity of the rabies virus infection, Rabipur can be administered to anyone who has been exposed to rabies, even pregnant women.
Severe allergic reactions (hypersensitivity)
If you or your child are known to be at risk of a severe allergic reaction to the vaccine or to any of its components, you or your child may be given a different rabies vaccine that does not contain such components. If no alternative vaccine is available, your doctor or nurse will discuss the risks of vaccination and rabies virus infection with you before you or your child receive the vaccine.
Warnings and precautions
In the case of an acute illness that requires treatment, vaccination is usually postponed until at least 2 weeks after recovery. The presence of a mild infection does not require postponing vaccination. However, consult your doctor or nurse first.
Consult your doctor or nurse before receiving Rabipur for post-exposure prophylaxis if you or your child:
- Have a severe allergy to egg or egg products (for symptoms, see section 4of this leaflet). Rabipur contains residual chicken protein from the manufacturing process.
- Have a severe allergy to the antibiotics neomycin, chlortetracycline, or amphotericin B. These antibiotics may be present in very small amounts in the vaccine.
- Have a severe allergy to polygelin.
Before or after any injection, fainting may occur, so you should inform your doctor or nurse if you or your child have fainted after receiving an injection in the past.
There have been reports of very rare but serious disorders affecting the nervous system after administration of the Rabipur vaccine. See section 4. Anti-inflammatory medications (steroids), which are often used to treat these disorders, may interfere with the effectiveness of the vaccine (see below, Use of Rabipur with other medicines). Your doctor or nurse will decide how to proceed in these circumstances.
Like all vaccines, Rabipur may not completely protect all people who are vaccinated.
The vaccine should not be administered in the buttocks, under the skin, or into a blood vessel.
Other medicines and Rabipur
Tell your doctor or nurse if you or your child are taking, have recently taken, or might take any other medicines, including those without a prescription. Unless your doctor tells you otherwise, you or your child should continue to take all prescribed medicines as usual.
If you or your child have a weakened immune system or are taking medicines that reduce the body's immunity to infections, it is still possible to administer Rabipur; however, you or your child may not be as well protected as other people. In this case, your doctor or your child's doctor may request blood tests after vaccination to check if the body has produced sufficient antibodies against the rabies virus. If necessary, you or your child will receive additional doses of the vaccine (see section 3of this leaflet).
Rabipurcan be administered at the same time as other inactivated vaccines.A different injection site will be used for each type of vaccine.
It may also be necessary for you or your child to receive an injection ofrabies antibodies(called “rabies immunoglobulin”) if you or your child have not been fully vaccinated against rabies and are likely to have been infected with the virus. In this case, you will be given the rabies immunoglobulin injection (which is given only onceand usually with the first dose of the vaccine) and the vaccine indifferent partsof the body.
Pregnancy and breastfeeding
If you are pregnant or breastfeeding, think you may be pregnant, or plan to become pregnant, you should still receive the rabies vaccine if you have had or are likely to have come into contact with the virus.
You can also be vaccinated with Rabipur during pregnancy or breastfeeding and before exposure to the virus, if it is thought that the risk of contact with the virus is considerable. In this case, your doctor will discuss the risks of vaccination and rabies virus infection with you and advise you on the best time to be vaccinated with Rabipur.
Driving and using machines
Some of the side effects listed in section 4of this leaflet may affect your ability to drive or use machines.
Rabipur contains sodium
This medicine contains less than 1mmol of sodium (23 mg) per dose, which is essentially “sodium-free”.
3. How to use Rabipur
Rabipur will be administered to you or your child by a doctor or nurse who has been trained to administer vaccines. Treatment that may be necessary to treat very severe allergic reactions that can occur after vaccination (see section 4of this leaflet) should be available. The vaccine should be administered to you or your child in a clinic or surgery that has the necessary equipment to treat these reactions.
Instructions for reconstitution of the vaccine for doctors and healthcare professionals are at the end of this leaflet.
The recommended dose for adults and children of any age is 1 milliliter (1.0 ml) per dose.
Your doctor will decide how many doses you or your child need, depending on whether you or your child have been vaccinated against rabies before or after any possible contact with the virus.
The vaccine is administered as an injection into a muscle (usually in the upper arm or, in young children, in the thigh muscle).
BEFORE ANY POSSIBLE CONTACT WITH THE VIRUS
If you or your child have never been vaccinatedagainst rabies:
- You need to receive 3 doses initially. The first dose is administered on the first visit; the second dose is administered 7 days later, and the third dose 21 or 28 days after the first dose.
- If you are an adult between 18 and 65 years of age who needs rapid protection, you can also receive three doses of Rabipur over 7 days. The first dose is administered on the first visit; the second dose is administered 3 days later, and the third dose 4 days after the second dose.
- Alternatively, if you are a person with a normal immune response, Rabipur can be administered in two doses over 7 days. The first dose is administered on the first visit, and the second dose 7 days later.
If you or your child miss a scheduled dose, you should make an appointment as soon as possible after the initially scheduled date.
The need for booster doses depends on the risk of contact with the rabies virus. Your doctor will consult official vaccination recommendations and inform you if a booster dose is necessary.
If you are at high risk of infection on an ongoing basis, your doctor may also ask you to have regular blood tests to determine the amount of rabies antibodies present in your blood, so that booster doses can be administered as soon as possible. Experience indicates that booster doses are usually needed every 2-5 years.
AFTER A SUSPECTED OR CONFIRMED CONTACT WITH THE VIRUS
People who have been vaccinated
If you or your child have been fully vaccinated against rabies and/or have received booster doses and have come into contact with an animal that has or is suspected to have rabies, you or your child will usually need 2 additional doses of the vaccine (each 1.0 ml). The first dose will be administered as soon as possible after contact, and the second dose 3 days later.
People who have not been vaccinated
If you or your child have not been vaccinated before or have received inadequate basic immunization, 4 or 5 doses (each 1.0 ml) will be administered, following one of the following schedules:
- If a 4-dose immunization schedule is used, the first 2 doses of the vaccine will be administered on day 0 as soon as possible after contact, and then single doses will be administered on days 7 and 21 after the first dose.
- An alternative 4-dose schedule can be used for healthy people who are known to have a good immune response; the first dose will be administered as soon as possible after contact on day 0, and the others on days 3, 7, and 14 after the first dose.
- If a 5-dose immunization schedule is used, the first dose of the vaccine will be administered on day 0 as soon as possible after contact, and the others on days 3, 7, 14, and 28 after the first dose.
After any possible contactwith the rabies virus, your doctor will assess the risk of infection based on the type of contact you or your child have had. For example, if you have been bitten or scratched by an animal that may have the virus, or have come into contact with bats, you will have a much higher risk of rabies virus infection than someone with intact skin who has been licked.
People with a weakened immune system (immunodeficiency)
If you or your child are at higher riskof rabies virus infection because your immune system is not working properly, you or your child will need 5 or 6 doses (each 1.0 ml) of rabies vaccine after contact with an animal that has or is suspected to have rabies. Vaccination will be administered in combination with local wound treatment and rabies immunoglobulin.
If 6 doses are used, the first 2 doses will be administered as soon as possible after contact, and then single doses will be administered on days 3, 7, 14, and 28 after the first dose.
If 5 doses are used, the first dose will be administered as soon as possible after contact, and the others on days 3, 7, 14, and 28 after the first dose.
You or your child may also need to have blood tests to measure the amount of rabies antibodies present in your blood, so that additional doses of the vaccine can be administered if necessary. Your doctor will explain the steps to follow and inform you when to come back for blood tests or additional doses.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
After vaccination with Rabipur, severe allergic reactions that affect the whole body may occur, sometimes associated with shock (dangerously low blood pressure)*. Adequate medical treatment and supervision should always be available in case of a rare severe allergic reaction to the vaccine. If this occurs, you should speak to a doctor immediately.
The most common side effects reported with the use of Rabipur were: pain at the injection site, mainly pain due to the injection, or hardening of the skin at the injection site. These reactions are very common (occurring in more than 1 in 10 people). Most injection site reactions were not severe and resolved within 24-48 hours after injection.
Other side effects include:
Very common(these may affect more than 1 in 10 people)
Headache
Dizziness
Rash
Malaise
Fatigue
Weakness
Fever
Common(these may affect up to 1 in 10 people)
Swollen glands
Loss of appetite
Nausea
Vomiting
Diarrhea
Abdominal pain
Hiccup
Muscle pain
Joint pain
Rare(these may affect up to 1 in 1,000 people)
Allergic reactions
Prickling or tingling sensation
Sweating
Chills
Very rare(these may affect up to 1 in 10,000 people)
Brain inflammation, nervous system disorders that can cause weakness, inability to move, or loss of sensation in certain parts of the body*
Fainting, unsteadiness with dizziness*
Severe allergic reaction that causes swelling of the face or throat*
*Adverse reactions reported spontaneously
Additional side effects in children
The frequency, type, and severity of adverse reactions in children are expected to be the same as in adults.
Reporting of side effects
If you or your child experience any side effects, talk to your doctor or nurse, even if they are not listed in this leaflet. You can also report side effects directly through the Spanish Medicines Agency's online reporting system, www.notificaRAM.es. By reporting side effects, you can help provide more information on the safety of this medicine.
5. Storage of Rabipur
Keep this vaccine out of the sight and reach of children.
Store in a refrigerator, protected from light (between 2 °C and 8 °C). Do not freeze.
Keep the vial and syringe in the outer packaging to protect them from light.
Do not use this vaccine after the expiry date stated on the original packaging. The expiry date is the last day of the month stated. Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. This will help protect the environment.
6. Container contents and additional information
Rabipur composition
The active ingredientof the vaccine is the rabies virus (inactivated, Flury LEP strain) ? 2.5 UI. This has been produced in purified chicken embryo cells (PCEC).
Other componentsare: trometamol, sodium chloride, disodium edetate, L-potassium glutamate, poligelina, sucrose, and water for injectable preparations.
It contains residues of chicken proteins (e.g., ovalbumin), human serum albumin, neomycin, chlortetracycline, and amphotericin B.
Appearance of the product and container contents
Rabipur is a white lyophilized powder that must be reconstituted with the clear and colorless solvent. The reconstituted vaccine is between transparent and slightly opalescent and between colorless and slightly pink.
Rabipur is supplied in containers containing 1 vial of powder, 1 pre-filled disposable syringe of sterile solvent with 2 identical needles (25 gauge, 25 mm), one for reconstitution and the other for injection.
Marketing authorization holder and manufacturer
Marketing authorization holder
Bavarian Nordic A/S
Philip Heymans Alle 3
2900 Hellerup
Denmark
Manufacturer
GSK Vaccines GmbH
Emil-von-Behring-Str. 76
35041 Marburg
Germany
or
Bavarian Nordic A/S
Hejreskovvej 10A
3490 Kvistgaard
Denmark
This medicinal product is authorized in the Member States of the European Economic Area under the following names:
Germany Rabipur
Austria Rabipur
Belgium Rabipur
Croatia Rabipur
Denmark Rabipur
Spain Rabipur
France Rabipur
Hungary Rabipur
Italy Rabipur
Luxembourg Rabipur
Norway Rabipur
Netherlands Rabipur
Poland Rabipur
Portugal Rabipur
Sweden Rabipur
Date of last revision of this leaflet: 11/2023
Detailed and updated information on this medicinal product is available on the website of the Spanish Agency for Medicines and Health Products (AEMPS) http://www.aemps.gob.es/.
Other sources of information
This information is intended only for healthcare professionals:
Instructions for use of the pre-filled disposable syringe of Rabipur
Pre-filled syringe
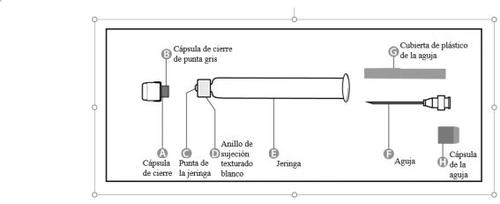
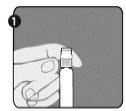
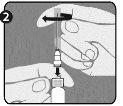
Step 1: Hold the syringe (E) with one hand, with the closure cap pointing upwards. Make sure to hold the syringe by the textured white retaining ring (D). |
|
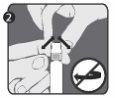
Step 2:Take the closure cap (A) with the other hand and tilt it firmly back and forth to break its connection with the retaining ring (D). Do not twist or bend the closure cap. |
|
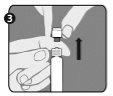
Step 3:Lift the closure cap (A) and the attached gray tip cap (B) to remove them. Be careful not to touch the sterile syringe tip (C). |
|
Application of the needle (these instructions are valid for the two supplied needles):
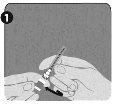
Step 1: Rotate the closure cap (H) of one of the two identical needles to remove it.This will be the needle used for reconstitution.Do not remove the plastic cover (G). |
|
Step 2:Hold the syringe (E) firmly with one hand by the textured white retaining ring (D). Insert the needle (F) with the other hand and rotate it clockwise until it clicks into place. Once the needle is locked, remove its plastic cover (G). The syringe (E) is now ready for use. |
|
Instructions for reconstituting Rabipur using the pre-filled syringe:
The vaccine must be inspected visually before and after reconstitution to detect any foreign particles and/or changes in physical appearance. The vaccine must not be used if any changes in its appearance have been detected. The reconstituted vaccine is between transparent and slightly opalescent and between colorless and slightly pink.
The powder for solution must be reconstituted with the supplied solvent for solution, gently shaking the mixture before injection. The reconstituted vaccine must be used immediately.
During manufacturing, the vial is sealed under vacuum. To avoid problems when withdrawing the reconstituted vaccine from the vial, it is recommended to unscrew the syringe from the needle after reconstitution of the vaccine to eliminate the negative pressure. After this, the vaccine can be easily withdrawn from the vial. It is not recommended to induce excessive pressure, as over-pressurization will create problems in extracting the adequate amount of vaccine.
The length of the needle is not sufficient to reach the bottom of the vial, so it is necessary to invert the vial and pull the needle back to bring it close to the stopper. This will allow the entire vaccine solution to be withdrawn from the vial.
After completing the reconstitution of the vaccine, remove the closure cap from the second needle (as explained in step 1 for the needles) and replace the needle used for reconstitution with the second needle, which will be used for administration. Do not use the same needle for reconstitution and administration.
-

- Country of registration
- Dosage formINJECTABLE, Greater than or equal to 2.5 IU
- ATC codeJ07BG01
- Active substance
- Prescription requiredYes
- Manufacturer
- This information is for reference only and does not constitute medical advice. Always consult a doctor before taking any medication. Oladoctor is not responsible for medical decisions based on this content.
- Alternatives to RABIPUR Powder and Solvent for Injectable Solution in Pre-filled SyringeDosage form: INJECTABLE, 3.25 IUActive substance: rabies, inactivated, whole virusManufacturer: Sanofi Winthrop IndustriePrescription requiredDosage form: INJECTABLE, 60 micrograms/dose + 60 micrograms/doseActive substance: respiratory syncytial virus vaccinesManufacturer: Pfizer Europe Ma EeigPrescription requiredDosage form: INJECTABLE, 3.75 microgramsActive substance: influenza, inactivated, split virus or surface antigenManufacturer: Glaxosmithkline BiologicalsPrescription required
Online doctors for RABIPUR Powder and Solvent for Injectable Solution in Pre-filled Syringe
Discuss questions about RABIPUR Powder and Solvent for Injectable Solution in Pre-filled Syringe, including use, safety considerations and prescription review, subject to medical assessment and local regulations.
Get RABIPUR Powder and Solvent for Injectable Solution in Pre-filled Syringe Prescription Online
Fill in a 2-min form
Tell us your symptoms, history, and what medicine you're requesting.
Pick a doctor or let us assign
Choose a specialist or we'll match you with the next available doctor.
Doctor reviews your case
Usually within 30 minutes. They may ask follow-up questions via chat.
Collect from any pharmacy
Electronic prescription sent to your email — valid across Spain.
Frequently Asked Questions
RABIPUR Powder and Solvent for Injectable Solution in Pre-filled Syringe requires a prescription in Spain. You can check with a doctor online whether this medicine may be appropriate for your situation.
The active ingredient in RABIPUR Powder and Solvent for Injectable Solution in Pre-filled Syringe is rabies, inactivated, whole virus. This information helps identify medicines with the same composition but different brand names.
RABIPUR Powder and Solvent for Injectable Solution in Pre-filled Syringe is manufactured by Bavarian Nordic A/S. Pharmacy brands and packaging may differ depending on the distributor.
Doctors such as Family doctors, Psychiatrists, Dermatologists, Cardiologists, Endocrinologists, Gastroenterologists, Pulmonologists, Nephrologists, Rheumatologists, Hematologists, Infectious disease physicians, Allergists, Geriatricians, Paediatricians, Oncologists may assess whether RABIPUR Powder and Solvent for Injectable Solution in Pre-filled Syringe is appropriate, depending on your situation and local regulations. You can book an online consultation to discuss your symptoms and possible next steps.
Spain has a well-developed healthcare infrastructure across major cities such as Madrid, Barcelona, Valencia, and Seville. Pharmacies are widely available and operate under strict regulations, ensuring access to prescribed medications.
You can buy RABIPUR Powder and Solvent for Injectable Solution in Pre-filled Syringe in Madrid, Barcelona, Seville, or Valencia at any local pharmacy with a valid prescription.
To obtain a prescription, you can use Oladoctor:
Other medicines with the same active substance (rabies, inactivated, whole virus) include VERORAB Powder and Solvent for Injectable Suspension in Pre-filled Syringe. Inactivated Rabies Vaccine, ABRYSVO powder and solvent for injectable solution, ADJUPANRIX 3.75 micrograms EMULSION AND SUSPENSION FOR INJECTABLE EMULSION. These may have different brand names or formulations but contain the same therapeutic ingredient. Always consult a doctor before switching or starting a new medicine.