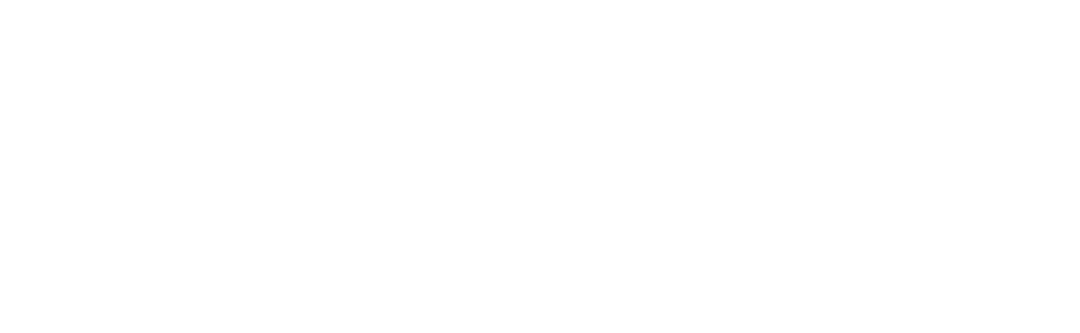
Numeta G13%e Preterm

Ask a doctor about a prescription for Numeta G13%e Preterm

How to use Numeta G13%e Preterm
Leaflet accompanying the packaging: information for the user
NUMETA G13%E Preterm, infusion emulsion
Please read the contents of the leaflet carefully before using the medicine, as it contains
important information for the patient.
- Please keep this leaflet, you may need to read it again.
- If you have any further questions, ask your child's doctor, pharmacist, or nurse.
- If your child experiences any side effects, including any not listed in this leaflet, please tell your doctor or nurse. See section 4.
Table of contents of the leaflet
- 1. What is NUMETA G13%E Preterm and what is it used for
- 2. Important information before giving NUMETA G13%E Preterm to your child
- 3. How to use NUMETA G13%E Preterm
- 4. Possible side effects
- 5. How to store NUMETA G13%E Preterm
- 6. Contents of the pack and other information
1. What is NUMETA G13%E Preterm and what is it used for
NUMETA G13%E Preterm is a specialized nutrition emulsion for premature infants. It is given through a tube inserted into a vein when the infant cannot take all the necessary nutrients by mouth.
The medicine comes in a three-chamber bag, where each chamber contains:
- 50% glucose solution;
- 5.9% pediatric amino acid solution with electrolytes;
- 12.5% fat emulsion.
Depending on the child's needs, two or three of these solutions are mixed in the bag before administration to the child.
NUMETA G13%E Preterm should only be used under medical supervision.
2. Important information before giving NUMETA G13%E Preterm to your child
When NUMETA G13%E Preterm should not be given to your child:
In the case of glucose and amino acid solutions mixed in the bag ("2 in 1"):
- NUMETA G13%E Preterm (or other calcium-containing solutions) must not be given at the same time as ceftriaxone (an antibiotic), even if separate infusion lines are used. There is a risk of solid particles forming in the bloodstream, which can be fatal.
In the case of solutions mixed with glucose, amino acids, and electrolytes, and fat emulsion in the bag ("3 in 1"):
In all the situations mentioned above for the "2 in 1" medicine, and additionally:
In all cases, the doctor will decide whether to give the medicine, taking into account the child's age, weight, and clinical condition. The doctor will also consider the results of all tests performed.
Warnings and precautions
Before starting NUMETA G13%E Preterm, discuss it with your child's doctor or nurse.
In the case of newborns and children under 2 years of age, the solution (in bags and administration sets) should be protected from light until the end of administration. Exposure of NUMETA G13%E Preterm to ambient light, especially after mixing with trace elements and/or vitamins, causes the formation of peroxides and other degradation products, which can be reduced by protecting from light.
Allergic reactions:
Infusion should be stopped immediately if any signs or symptoms of an allergic reaction occur (such as fever, sweating, chills, headache, skin rash, or difficulty breathing). This medicine contains soybean oil, which can rarely cause hypersensitivity reactions. In some people allergic to peanut proteins, soy protein allergy has also been observed. NUMETA G13%E Preterm contains glucose produced from cornstarch. Therefore, NUMETA G13%E Preterm should be used with caution in patients with known allergy to corn or corn-containing products.
Risk of solid particle formation with ceftriaxone (antibiotic):
The antibiotic ceftriaxone should not be mixed or given at the same time as any calcium-containing solutions (including NUMETA G13%E Preterm) by intravenous infusion. The doctor is aware of this and will not give them to your child at the same time, even through separate infusion lines or different infusion sites.
Formation of small particles in the blood vessels of the lungs:
Breathing difficulties can also be a sign that small particles have formed, blocking the blood vessels in the lungs (pulmonary vascular deposits). If your child experiences any breathing difficulties, please tell your doctor or nurse. They will decide on the appropriate action.
Infection and sepsis:
The doctor will carefully monitor your child to detect signs of infection. Following aseptic procedures (procedures that protect against microorganisms) when inserting and maintaining the catheter and when preparing the nutritional mixture can reduce the risk of infection.
Sometimes, if a tube is inserted into a vein (central venous catheter), your child may develop an infection and sepsis (presence of bacteria in the blood). Some medicines and diseases can increase the risk of infection or sepsis. Patients requiring parenteral nutrition (feeding through a tube inserted into a vein) due to their health condition may be more prone to developing infections.
Fat overload syndrome:
During the use of similar medicines, the occurrence of fat overload syndrome has been reported. Reduced or impaired ability to eliminate fats contained in NUMETA G13%E Preterm or overdose can result in so-called fat overload syndrome (see sections 3 and 4).
Changes in blood chemical composition:
The doctor will check and monitor your child's fluid status, blood chemical composition, and the content of other substances during treatment with NUMETA G13%E Preterm. Sometimes, feeding severely malnourished patients can cause significant changes in blood chemical composition, which may require correction. Additional fluid may also accumulate in tissues and cause swelling. It is recommended to start parenteral nutrition slowly and cautiously.
Monitoring and adjustment:
The doctor will carefully monitor and adjust the administration of NUMETA G13%E Preterm according to your child's individual needs, especially in the following situations:
- severe trauma;
- severe diabetes;
- shock;
- heart attack;
- severe infection;
- certain types of coma.
Use with caution:
NUMETA G13%E Preterm should be used with caution if your child has:
- pulmonary edema (fluid in the lungs) or heart failure;
- severe liver dysfunction;
- malabsorption;
- high blood sugar levels;
- kidney problems;
- severe metabolic disorders (when the breakdown process of substances does not proceed normally);
- bleeding disorders.
Your child's fluid status, liver function tests, and/or other blood test results will be carefully checked.
Information on the use of this medicine in premature infants born before 28 weeks of gestation is limited.
NUMETA G13%E Preterm and other medicines
Tell your doctor about all medicines your child is currently taking or has recently taken, as well as any medicines that will be taken.
NUMETA G13%E Preterm must not be given at the same time as:
- ceftriaxone(antibiotic), even in separate infusion lines, due to the risk of solid particle formation;
- bloodthrough the same infusion set, due to the risk of pseudoagglutination (red blood cells clumping);
- ampicillin, phenytoin, or furosemidethrough the same infusion line, due to the risk of solid particle formation.
Warfarin and coumarin (anticoagulants):
The doctor will carefully monitor your child if warfarin or coumarin is being used. These medicines are anticoagulants used to prevent blood clotting. Vitamin K1 is a natural component of olive oil and soybean oil. Vitamin K1 may interact with medicines such as warfarin and coumarin.
Lab tests:
The lipids in this emulsion may affect the results of certain laboratory tests. Laboratory tests may be performed 5 to 6 hours after the administration of lipids or after the last administration.
Interactions between NUMETA G13%E Preterm and medicines that may affect potassium levels/metabolism:
NUMETA G13%E Preterm contains potassium. High levels of potassium in the blood can cause abnormal heart rhythms. Patients taking diuretics (medicines that reduce fluid retention) or ACE inhibitors (medicines used for high blood pressure) or angiotensin II receptor antagonists (medicines used for high blood pressure) or immunosuppressive medicines (medicines that can reduce the normal immune response of the body) should be closely monitored. These types of medicines can increase potassium levels.
3. How to use NUMETA G13%E Preterm
NUMETA G13%E Preterm should always be given to your child according to the doctor's instructions.
In case of doubts, please contact your child's doctor.
Age group
NUMETA G13%E Preterm is designed to meet the nutritional needs of premature infants.
NUMETA G13%E Preterm may not be suitable for some premature infants, as their condition may require individually tailored nutrition to meet their specific needs. The doctor will decide whether this medicine is suitable for your child.
Administration
This medicine is an infusion emulsion. It is given through a plastic tube into a vein in the arm or a large vein in the chest.
The doctor may decide not to give your child lipids. The NUMETA G13%E Preterm bag is designed so that, if necessary, the partitions can be broken only between the amino acid/electrolyte chamber and the glucose chamber. In this case, the partition between the amino acid chamber and the lipid chamber remains intact. This way, the contents of the bag can be administered without lipids.
In the case of newborns and children under 2 years of age, the solution (in bags and administration sets) should be protected from light until the end of administration (see section 2).
Dose and duration of treatment
The doctor will decide on the dose of the medicine and how long it will be given. The dose depends on your child's nutritional needs. The dose will be determined based on your child's weight, clinical condition, and ability to break down and use the components of NUMETA G13%E Preterm.
Additional nutritional components or proteins can also be given orally/enterally.
Use of NUMETA G13%E Preterm in children older than the recommended age
Symptoms
Too high a dose of the medicine or too rapid administration can cause:
- nausea (vomiting);
- vomiting;
- seizures;
- electrolyte disturbances (abnormal amounts of electrolytes in the blood);
- symptoms of hypervolemia (increased circulating blood volume, excess fluid in the blood vessels);
- acidosis (increased blood acidity). In such cases, the infusion should be stopped immediately. The doctor will decide whether additional actions are required.
Overdose of fats contained in NUMETA G13%E Preterm can lead to the development of "fat overload syndrome", which usually resolves after the infusion is stopped. In newborns (infants) and young children (children under 2 years of age), fat overload syndrome has been associated with respiratory distress leading to decreased oxygen levels in the body (respiratory failure) and conditions leading to increased blood acidity (acidosis). To prevent such situations, the doctor will regularly check your child's condition and perform blood tests during treatment.
4. Possible side effects
Like all medicines, NUMETA G13%E Preterm can cause side effects, although not all children will experience them.
If you notice any changes in your child's condition during or after treatment, please tell your doctor or nurse immediately.
The tests performed by the doctor during the use of the medicine by your child should minimize the risk of side effects.
If symptoms of an allergic reaction occur, the infusion should be stopped and your doctor or nurse should be contacted immediately. This can be a serious reaction, and symptoms may include:
- sweating
- chills
- headache
- skin rash
- difficulty breathing
Other observed side effects:
Common: may affect up to 1 in 10 people
- Low phosphate levels in the blood (hypophosphatemia)
- High blood sugar levels (hyperglycemia)
- High calcium levels in the blood (hypercalcemia)
- High triglyceride levels in the blood (hypertriglyceridemia)
- Electrolyte disturbances (hyponatremia)
Uncommon: may affect up to 1 in 100 people
- High lipid levels in the blood (hyperlipidemia)
- A condition in which bile cannot flow from the liver to the duodenum (cholestasis). The duodenum is a part of the intestine.
Rare: cannot be estimated from the available data(These side effects have been reported only after peripheral administration of NUMETA G13%E Preterm and NUMETA G16%E with inadequate dilution).
- Necrosis of the skin
- Soft tissue damage
- Extravasation
The following side effects have been reported for other parenteral nutrition medicines:
- Reduced or impaired ability to eliminate lipids contained in NUMETA G13%E Preterm can lead to the development of fat overload syndrome. The following signs and symptoms of this syndrome usually resolve after the infusion of the fat emulsion is stopped:
- Sudden and severe deterioration of the patient's clinical condition
- High levels of fat in the blood (hyperlipidemia)
- Fever
- Fatty liver (hepatomegaly)
- Impaired liver function
- Decreased red blood cell count, which can cause pale skin and be the cause of weakness or shortness of breath (anemia)
- Decreased white blood cell count, which can increase the risk of infection (leukopenia)
- Decreased platelet count, which can increase the risk of bruising and/or bleeding (thrombocytopenia)
- Blood clotting disorders, which affect the ability of blood to form clots
- Respiratory distress leading to decreased oxygen levels in the body (respiratory failure)
- Conditions leading to increased blood acidity (acidosis)
- Coma requiring hospitalization
- Formation of small particles that can block blood vessels in the lungs (pulmonary vascular deposits) or cause breathing difficulties.
Reporting side effects
If your child experiences any side effects, please tell your doctor or nurse.
This includes any side effects not listed in this leaflet.
Side effects can be reported directly to the Department of Adverse Reaction Monitoring of Medicinal Products, Medical Devices, and Biocidal Products
Al. Jerozolimskie 181 C
PL 02-222 Warsaw
Tel.: +48 22 49 21 301
Fax: +48 22 49 21 309
Website: https://smz.ezdrowie.gov.pl
Side effects can also be reported to the marketing authorization holder.
By reporting side effects, you can help provide more information on the safety of this medicine.
5. How to store NUMETA G13%E Preterm
Keep the medicine out of the sight and reach of children when not in use.
In the case of newborns and children under 2 years of age, the solution (in bags and administration sets) should be protected from light until the end of administration (see section 2).
Do not use this medicine after the expiry date stated on the bag and outer packaging (MM/RRRR). The expiry date refers to the last day of the specified month.
Do not freeze.
Store in the protective bag.
Medicines should not be disposed of via wastewater or household waste. Please ask your pharmacist how to dispose of medicines no longer required. These measures will help protect the environment.
6. Contents of the pack and other information
What NUMETA G13%E Preterm looks like and what the pack contains
NUMETA G13%E Preterm is contained in a three-chamber bag. The contents of each bag are sterile and consist of a glucose solution, an amino acid solution for children with electrolytes, and a fat emulsion, as described below.
| Container size | 50% glucose solution | 5.9% amino acid solution with electrolytes | 12.5% fat emulsion |
| 300 ml | 80 ml | 160 ml | 60 ml |
Appearance before preparation:
- The solutions in the chambers containing amino acids and glucose are clear, colorless, or slightly yellow
- The fat emulsion is a uniform, milky-white liquid
Appearance after preparation:
- The "2 in 1" infusion solutions (amino acids/electrolytes and glucose) are clear, colorless, or slightly yellow
- The "3 in 1" infusion emulsion is uniform and milky-white
The three-chamber bag is a multi-layer plastic bag.
To protect it from air, the NUMETA G13%E Preterm bag is packaged in a protective bag that protects against oxygen, which also contains an oxygen absorber and an oxygen indicator.
Package sizes
300 ml bags: 10 bags in a cardboard box
1 bag of 300 ml
Not all pack sizes may be marketed.
Marketing authorization holder and manufacturer
Marketing authorization holder
Baxter Polska Sp. z o.o.
ul. Kruczkowskiego 8
00-380 Warsaw
Manufacturer
Baxter S.A.
Boulevard René Branquart 80
7860 Lessines
Belgium
This medicinal product is authorized in the Member States of the European Economic Area under the following names:
| Country | Name |
| Austria Germany | Numeta G 13 % E Emulsion zur Infusion |
| Belgium Luxembourg | NUMETZAH G13%E, émulsion pour perfusion |
| France | NUMETAH G13%E PREMATURES, emulsion pour perfusion |
| Denmark Norway Sweden | Numeta G13E |
| Czech Republic | NUMETA G 13 % E |
| Greece | NUMETA Preterm G 13 E |
| Netherlands | NUMETA G13%E emulsie voor infusie |
| Ireland Malta United Kingdom | Numeta G13%E Preterm, Emulsion for Infusion |
| Italy | NUMETA G13E emulsione per infusione |
| Finland | Numeta G13E infuusioneste, emulsio |
| Poland | NUMETA G13%E Preterm |
| Portugal | Numeta G13%E |
| Spain | NUMETA G13%E, emulsión para perfusión |
Date of last revision of the leaflet: May 2024
Information intended for healthcare professionals only
*In some cases, this medicine may be administered at home by parents or other caregivers.
In these cases, parents/caregivers should read the following information.
Do not add other ingredients to the bag without first checking their compatibility.
This could cause the formation of solid particles or destabilization of the fat emulsion. This can lead to blockage of blood vessels.
NUMETA G13%E Preterm should reach room temperature before use.
Before using NUMETA G13%E Preterm, prepare the bag as shown below.
Make sure the bag is not damaged. The bag can only be used if it is not damaged. The following are the characteristics of an undamaged bag:
- The partitions are intact. This is indicated by the lack of mixing of the contents of any of the three chambers.
- The amino acid and glucose solutions are clear, colorless, or slightly yellow, without visible particles.
- The fat emulsion is a uniform, milky-white liquid.
Before opening the protective bag, check the color of the oxygen indicator.
- Compare it with the reference color printed next to the OK symbol and shown on the labeled area of the oxygen indicator.
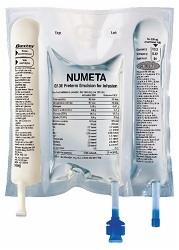
- Do not use the medicine if the color of the oxygen indicator does not match the reference color printed next to the OK symbol. Figures 1 and 2 show how to remove the protective bag. Discard the protective bag, oxygen indicator, and oxygen absorber.









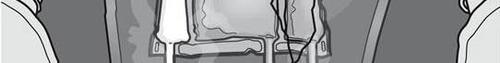
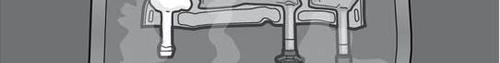




Figure 1
Figure 2
Preparing the mixed emulsion:
- Before breaking the seals, make sure the medicine has reached room temperature.
- Place the bag on a flat, clean surface.
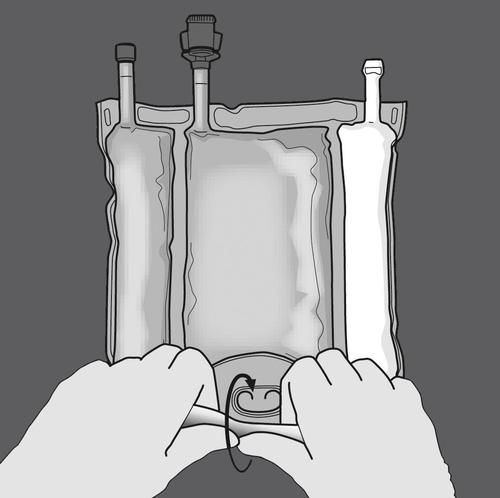
Activating the three-chamber bag (mixing 3 solutions by opening 2 breakable seals)
Step 1: Start rolling the bag from the side with the hanger.
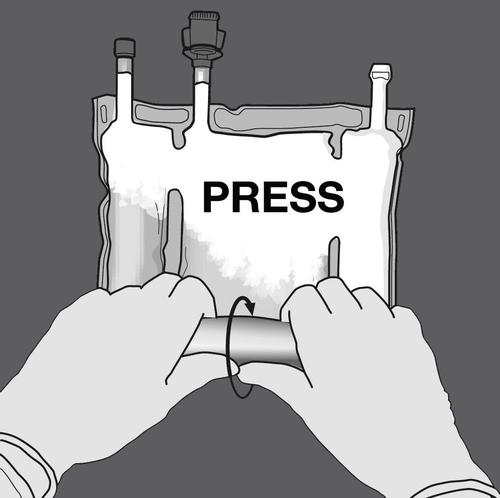
Step 2: Press until the seals open.
PRESS
Step 3: Change the direction by rolling the bag towards the hanger. Continue until the seal is completely open.
Repeat the same procedure to completely open the second seal.
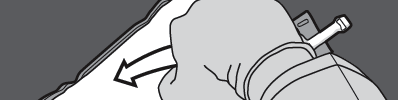
PRESS
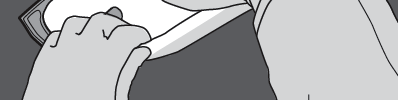

Step 4: Turn the bag at least three times to mix the contents thoroughly.
The mixed solution should have a milky-white emulsion appearance.

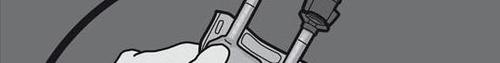






Step 5: Remove the protective plug from the administration port and insert the infusion set.








Activating the two-chamber bag (mixing 2 solutions by opening the breakable seal between the chambers)
Step 1: To mix only 2 solutions, roll the bag starting from the top corner (with the hanger) next to the seal separating the glucose and amino acid solutions.
Press to open the seal separating the amino acid and glucose solutions.
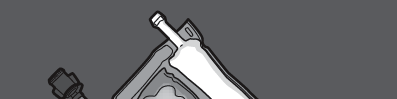
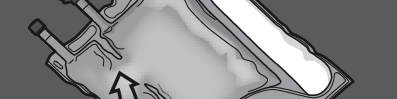
PRESS

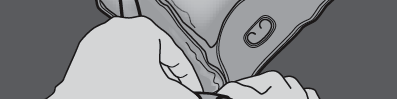
Step 2: Place the bag so that the chamber with the fat emulsion is closest to you.
Roll the bag, protecting the chamber with the fat emulsion with your hands.
Step 3: Press with one hand and roll the bag towards the tubes.

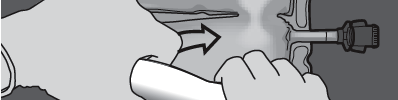
PRESS


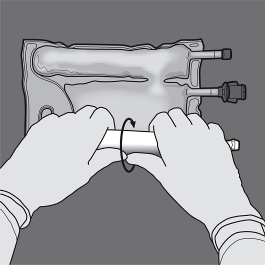
Step 4: Change the direction by rolling the bag towards the top end (with the hanger).
Press with your second hand until the seal separating the amino acid and glucose solutions is completely open.

PRESS


Step 5: Turn the bag at least three times to mix the contents thoroughly.
The mixed solution should be clear, colorless or slightly yellow.







Step 6: Remove the protective cap from the administration site and introduce the infusion set.








Within the first hour, the flow rate should be gradually increased. The infusion rate must be adjusted based on the following factors:
- administered dose;
- daily volume intake;
- infusion duration.
Administration method:
In the case of use in newborns and children under 2 years of age, the solution (in bags and infusion sets) should be protected from light until the end of administration.
For the administration of NUMETA G13%E Preterm, it is recommended to use a 1.2 micron filter.
Due to the high osmolality, the undiluted NUMETA G13%E Preterm can only be administered through a central vein. Appropriate dilution of NUMETA G13%E Preterm with water for injection reduces osmolality and allows infusion into a peripheral vessel. The following formula shows the effect of dilution on the osmolality of the solution.
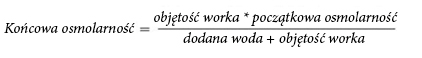
In the table below, examples of osmolality of activated dual-chamber and triple-chamber bags after adding water for injection are presented:
| Amino acids and glucose (activated W2K) | Amino acids, glucose and lipids (activated W3K) | |
| Initial volume in the bag (ml) | 240 | 300 |
| Initial osmolality (mOsm/l approximately) | 1400 | 1150 |
| Volume of added water (ml) | 240 | 300 |
| Final volume after addition (ml) | 480 | 600 |
| Osmolality after addition (mOsm/l approximately) | 700 | 575 |
Introduction of additional components:
Exposure of parenteral nutrition solutions to light, especially after mixing with trace elements and/or vitamins, may have an adverse effect on clinical outcomes in newborns, due to the production of peroxides and other degradation products. In the case of use in newborns and children under 2 years of age, NUMETA G13%E Preterm should be protected from environmental light until the end of administration.
Compatible additional components can be added to the reconstituted mixture through the injection site (after opening the breakable seams and mixing the contents of two or three chambers).
Vitamins can also be added to the glucose chamber before reconstituting the mixture (before opening the breakable seams and before mixing the solutions and emulsions).
Possible additions of ready-to-use solutions of trace elements (identified as TE1 and TE4), vitamins (identified as lyophilized V1 and emulsion V2) and electrolytes in specified amounts are presented in Tables 1-4.
- 1.Compatibility with TE4, V1 and V2
Table 1: Compatibility of 3-in-1 (activated W3K) with water dilution and without dilution
| For 300 ml (after mixing 3 chambers, with lipids) | ||||||
| Addition without dilution | Addition with dilution | |||||
| Additional components | Contained amount | Maximum added amount | Maximum total amount | Contained amount | Maximum added amount | Maximum total amount |
| Sodium (mmol) | 6,6 | 5,0 | 11,6 | 6,6 | 5,0 | 11,6 |
| Potassium (mmol) | 6,2 | 4,2 | 10,4 | 6,2 | 4,2 | 10,4 |
| Magnesium (mmol) | 0,47 | 0,83 | 1,3 | 0,47 | 0,83 | 1,3 |
| Calcium (mmol) | 3,8 | 3,5 | 7,3 | 3,8 | 3,5 | 7,3 |
| Phosphates* (mmol) | 3,8 | 2,5 | 6,3 | 3,8 | 2,5 | 6,3 |
| Trace elements and vitamins | 15 ml TE4 + 1,5 vials V1 + 25 ml V2 | 15 ml TE4 + 1,5 vials V1 + 25 ml V2 | 15 ml TE4 + 1,5 vials V1 + 25 ml V2 | 15 ml TE4 + 1,5 vials V1 + 25 ml V2 | ||
| Water for injection | 300 ml | 300 ml | ||||
Table 2: Compatibility of 2-in-1 (activated W2K) with water dilution and without dilution
| For 240 ml (after mixing 2 chambers, without lipids) | ||||||
| Addition without dilution | Addition with dilution | |||||
| Additional components | Contained amount | Maximum added amount | Maximum total amount | Contained amount | Maximum added amount | Maximum total amount |
| Sodium (mmol) | 6,4 | 17,6 | 24 | 6,4 | 0,0 | 6,4 |
| Potassium (mmol) | 6,2 | 17,8 | 24 | 6,2 | 0,0 | 6,2 |
| Magnesium (mmol) | 0,47 | 2,13 | 2,6 | 0,47 | 0,0 | 0,47 |
| Calcium (mmol) | 3,8 | 3,5 | 7,3 | 3,8 | 0,0 | 3,8 |
| Phosphates* (mmol) | 3,2 | 4,0 | 7,2 | 3,2 | 0,0 | 3,2 |
| Trace elements and vitamins | 2,5 ml TE4 + ¼ vial V1 | 2,5 ml TE4 + ¼ vial V1 | 2,5 ml TE4 + ¼ vial V1 | 2,5 ml TE4 + ¼ vial V1 | ||
| Water for injection | 240 ml | 240 ml |
- 2.Compatibility with TE1, V1 and V2Table 3: Compatibility of 3-in-1 (activated W3K) with water dilution and without dilution
| For 300 ml (after mixing 3 chambers, with lipids) | ||||||
| Addition without dilution | Addition with dilution | |||||
| Additional components | Contained amount | Maximum added amount | Maximum total amount | Contained amount | Maximum added amount | Maximum total amount |
| Sodium (mmol) | 6,6 | 5,0 | 11,6 | 6,6 | 0,0 | 6,6 |
| Potassium (mmol) | 6,2 | 4,2 | 10,4 | 6,2 | 0,0 | 6,2 |
| Magnesium (mmol) | 0,47 | 0,83 | 1,3 | 0,47 | 0,0 | 0,47 |
| Calcium (mmol) | 3,8 | 1,9 | 5,7 | 3,8 | 0,0 | 3,8 |
| Phosphates* (mmol) | 3,8 | 2,5 | 6,3 | 3,8 | 0,0 | 3,8 |
| Trace elements and vitamins | 2,5 ml TE1 + ¼ vial V1 + 2,5 ml V2 | 2,5 ml TE1 + ¼ vial V1 + 2,5 ml V2 | 2,5 ml TE1 + ¼ vial V1 + 2,5 ml V2 | 2,5 ml TE1 + ¼ vial V1 + 2,5 ml V2 | ||
| Water for injection | 300 ml | 300 ml | ||||
Table 4: Compatibility of 2-in-1 (activated W2K) with water dilution and without dilution
| For 240 ml (after mixing 2 chambers, without lipids) | ||||||
| Addition without dilution | Addition with dilution | |||||
| Additional components | Contained amount | Maximum added amount | Maximum total amount | Contained amount | Maximum added amount | Maximum total amount |
| Sodium (mmol) | 6,4 | 17,6 | 24 | 6,4 | 0,0 | 6,4 |
| Potassium (mmol) | 6,2 | 17,8 | 24 | 6,2 | 0,0 | 6,2 |
| Magnesium (mmol) | 0,47 | 2,13 | 2,6 | 0,47 | 0,0 | 0,47 |
| Calcium (mmol) | 3,8 | 3,5 | 7,3 | 3,8 | 0,0 | 3,8 |
| Phosphates* (mmol) | 3,2 | 4,0 | 7,2 | 3,2 | 0,0 | 3,2 |
| Trace elements and vitamins | 2,5 ml TE1 + ¼ vial V1 | 2,5 ml TE1 + ¼ vial V1 | 2,5 ml TE1 + ¼ vial V1 | 2,5 ml TE1 + ¼ vial V1 | ||
| Water for injection | 240 ml | 240 ml | ||||
The composition of preparations containing vitamins and trace elements is presented in Tables 5 and 6.
Table 5: Composition of the used ready-to-use preparation containing trace elements:
Table 6: Composition of the used ready-to-use preparation containing vitamins:
Introduction of additional components:
- Aseptic conditions must be maintained.
- Prepare the injection site on the bag.
- Puncture the injection site and inject additional components using a syringe needle or a device for preparing the drug.
- Mix the contents of the bag with the additional components.
Preparation of infusion:
- Aseptic conditions must be maintained.
- Suspend the bag.
- Remove the plastic protector from the administration port.
- Insert the infusion set needle into the administration port with a firm motion.
Infusion administration:
- For single use only.
- Administer the drug only after breaking the seams between the two or three chambers and mixing the contents of the two or three chambers.
- Ensure that the activated triple-chamber bag infusion emulsion does not separate phases or that the activated dual-chamber bag infusion solution does not contain any solid particles.
- It is recommended to use immediately after opening the breakable seams. NUMETA G13%E Preterm should not be stored for the next infusion.
- Do not connect partially used bags.
| Composition per vial | TE1 (10 ml) | TE4 (10 ml) |
| Zinc | 38,2 µmol or 2,5 mg | 15,3 µmol or 1 mg |
| Selenium | 0,253 µmol or 0,02 mg | 0,253 µmol or 0,02 mg |
| Copper | 3,15 µmol or 0,2 mg | 3,15 µmol or 0,2 mg |
| Iodine | 0,0788 µmol or 0,01 mg | 0,079 µmol or 0,01 mg |
| Fluorine | 30 µmol or 0,57 mg | |
| Manganese | 0,182 µmol or 0,01 mg | 0,091 µmol or 0,005 mg |
| Composition per vial | V1 | V2 |
| Vitamin B1 | 2,5 mg | |
| Vitamin B2 | 3,6 mg | |
| Niacinamide | 40 mg | |
| Vitamin B6 | 4,0 mg | |
| Pantothenic acid | 15,0 mg | |
| Biotin | 60 µg | |
| Folic acid | 400 µg | |
| Vitamin B12 | 5,0 µg | |
| Vitamin C | 100 mg | |
| Vitamin A | 2300 IU | |
| Vitamin D | 400 IU | |
| Vitamin E | 7 IU | |
| Vitamin K | 200 µg |
- To avoid the formation of an air embolism caused by residual air in the first bag, do not connect bags in series.
- For the administration of NUMETA G13%E Preterm, it is recommended to use a 1.2 micron filter.
- In the case of use in newborns and children under 2 years of age, protect from light until the end of administration. Exposure of NUMETA G13%E Preterm to environmental light, especially after mixing with trace elements and/or vitamins, causes the production of peroxides and other degradation products, which can be reduced by protecting from light.
- Any unused medicinal product or waste material should be disposed of in accordance with local requirements.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help protect the environment.
What NUMETA G13%E Preterm contains
Active substances:
| Active substance | Activated W2K (240 ml) | Activated W3K (300 ml) |
| Amino acid chamber | ||
| Alanine | 0,75 g | 0,75 g |
| Arginine | 0,78 g | 0,78 g |
| Aspartic acid | 0,56 g | 0,56 g |
| Cysteine | 0,18 g | 0,18 g |
| Glutamic acid | 0,93 g | 0,93 g |
| Glycine | 0,37 g | 0,37 g |
| Histidine | 0,35 g | 0,35 g |
| Isoleucine | 0,62 g | 0,62 g |
| Leucine | 0,93 g | 0,93 g |
| Lysine monohydrate (corresponding to lysine) | 1,15 g (1,03 g) | 1,15 g (1,03 g) |
| Methionine | 0,22 g | 0,22 g |
| Ornithine hydrochloride (corresponding to ornithine) | 0,30 g (0,23 g) | 0,30 g (0,23 g) |
| Phenylalanine | 0,39 g | 0,39 g |
| Proline | 0,28 g | 0,28 g |
| Serine | 0,37 g | 0,37 g |
| Taurine | 0,06 g | 0,06 g |
| Threonine | 0,35 g | 0,35 g |
| Tryptophan | 0,19 g | 0,19 g |
| Tyrosine | 0,07 g | 0,07 g |
| Valine | 0,71 g | 0,71 g |
| Potassium acetate | 0,61 g | 0,61 g |
| Calcium chloride dihydrate | 0,55 g | 0,55 g |
| Magnesium acetate tetrahydrate | 0,10 g | 0,10 g |
| Sodium glycerophosphate hydrate | 0,98 g | 0,98 g |
| Glucose chamber | ||
| Glucose monohydrate (corresponding to anhydrous glucose) | 44,00 g (40,00 g) | 44,00 g (40,00 g) |
| Lipid chamber | ||
| Purified olive oil (approx. 80%) + purified soybean oil (approx. 20%) | 7,5 g | |
The solution/emulsion after mixing contains the following components:
| Composition | ||||
| Activated W2K | Activated W3K | |||
| Per unit of volume (ml) Nitrogen (g) Amino acids (g) Glucose (g) Lipids (g) Energy value Total energy value (kcal) | 240 1,4 9,4 40,0 0 198 | 100 0,59 3,9 16,7 0 82 | 300 1,4 9,4 40,0 7,5 273 | 100 0,47 3,1 13,3 2,5 91 |
| Non-protein energy value (kcal) Glucose energy value (kcal) Lipid energy value (kcal) Non-protein energy/nitrogen (kcal/g N) Lipid energy/non-protein energy (%) Lipid energy/total energy (%) Electrolytes Sodium (mmol) Potassium (mmol) Magnesium (mmol) Calcium (mmol) Phosphatesb (mmol) Acetates (mmol) Malates (mmol) Chlorides (mmol) | 160 160 0 113 Not applicable Not applicable 6,4 6,2 0,47 3,8 3,2 7,2 3,2 9,3 | 67 67 0 113 Not applicable Not applicable 2,7 2,6 0,20 1,6 1,3 3,0 1,3 3,9 | 235 160 75 165 32 28 6,6 6,2 0,47 3,8 3,8 7,2 3,2 9,3 | 78 53 25 165 32 28 2,2 2,1 0,16 1,3 1,3 2,4 1,1 3,1 |
| pH (approximately) Osmolality (approximately) (mOsm/l) | 5,5 1400 | 5,5 1400 | 5,5 1150 | 5,5 1150 |
This includes the energy value of phospholipids from egg yolk for injection.
This includes phosphates from phospholipids from egg yolk for injection, which are a component of the lipid emulsion.
Other ingredients:
L-malic acid
Hydrochloric acid
Egg yolk phospholipids for injection
Glycerol
Sodium oleate
Sodium hydroxide
Water for injection
to adjust pH
Baxter, Numeta, Numetzah and Numetah are trademarks of Baxter International Inc.
- Country of registration
- Active substance
- Prescription requiredYes
- Manufacturer
- ImporterBaxter S.A.
- This information is for reference only and does not constitute medical advice. Always consult a licensed doctor before taking any medication. Oladoctor is not responsible for medical decisions based on this content.
- Alternatives to Numeta G13%e PretermDosage form: Solution, -Active substance: combinationsPrescription not requiredDosage form: Solution, -Active substance: combinationsPrescription not requiredDosage form: Solution, -Active substance: combinationsPrescription not required
Alternatives to Numeta G13%e Preterm in other countries
The best alternatives with the same active ingredient and therapeutic effect.
Alternative to Numeta G13%e Preterm in Spain
Alternative to Numeta G13%e Preterm in Ukraine
Online doctors for Numeta G13%e Preterm
Discuss dosage, side effects, interactions, contraindications, and prescription renewal for Numeta G13%e Preterm – subject to medical assessment and local rules.